
Concept explainers
(a)
Interpretation: The condensed formula of the given molecule is to be converted to Lewis structure.
Concept introduction: Lewis structures can be drawn for organic compounds which contain both rings and chains of atoms. In Lewis structure compounds are represented with symbols of atoms connected with bonds.
Answer to Problem 1.65P
The Lewis structure of the given molecule is shown as,
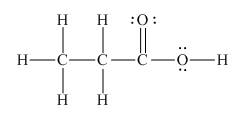
Figure 1
Explanation of Solution
Lewis structures can be drawn by following the below rules.
• Draw a carbon back bone of the compound.
• Each carbon atom has enough hydrogens to make it tetravalent.
• All heteroatoms and the hydrogens directly bonded to them are drawn.
To interpret a condensed formula, one should start from left and proceed to right. All the carbon atoms are tetravalent. A carbon bonded to three

Figure 1
The Lewis structure of the given molecule is shown in Figure 1.
(b)
Interpretation: The condensed formula of the given molecule is to be converted to Lewis structure.
Concept introduction: Lewis structures can be drawn for organic compounds which contains both rings and chains of atoms. In Lewis structure compounds are represented with symbols of atoms connected with bonds.
Answer to Problem 1.65P
The Lewis structure of the given molecule is shown as, is,
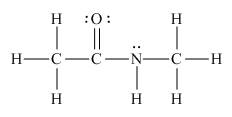
Figure 2
Explanation of Solution
Lewis structures can be drawn by following the below rules.
• Draw a carbon back bone of the compound.
• Each carbon atom has enough hydrogens to make it tetravalent.
• All heteroatoms and the hydrogens directly bonded to them are drawn.
To interpret a condensed formula, one should start from left and proceed to right. All the carbon atoms are tetravalent. A carbon bonded to three

Figure 2
The Lewis structure of the given molecule is shown in Figure 2.
(c)
Interpretation: The condensed formula of the given molecule is to be converted to Lewis structure.
Concept introduction: Lewis structures can be drawn for organic compounds which contains both rings and chains of atoms. In Lewis structure compounds are represented with symbols of atoms connected with bonds.
Answer to Problem 1.65P
The Lewis structure of the given molecule is shown as,
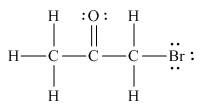
Figure 3
Explanation of Solution
Lewis structures can be drawn by following the below rules.
• Draw a carbon back bone of the compound.
• Each carbon atom has enough hydrogens to make it tetravalent.
• All heteroatoms and the hydrogens directly bonded to them are drawn.
To interpret a condensed formula, one should start from left and proceed to right. All the carbon atoms are tetravalent. A carbon bonded to three

Figure 3
The Lewis structure of the given molecule is shown in Figure 3.
(d)
Interpretation: Condensed formula of the given molecule is to be converted to Lewis structure.
Concept introduction: Lewis structures can be drawn for organic compounds which contains both rings and chains of atoms. In Lewis structure compounds are represented with symbols of atoms connected with bonds.
Answer to Problem 1.65P
The Lewis structure of the given molecule is,
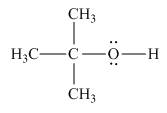
Figure 4
Explanation of Solution
Lewis structures can be drawn by following the below rules.
• Draw a carbon back bone of the compound.
• Each carbon atom has enough hydrogens to make it tetravalent.
• All heteroatoms and the hydrogens directly bonded to them are drawn.
To interpret a condensed formula one should start from left and proceed to right. All the carbon atoms are tetravalent. A carbon bonded to three

Figure 4
The Lewis structure of the given molecule is shown in Figure 4.
(e)
Interpretation: Condensed formula of the given molecule is to be converted to Lewis structure.
Concept introduction: Lewis structures can be drawn for organic compounds which contains both rings and chains of atoms. In Lewis structure compounds are represented with symbols of atoms connected with bonds.
Answer to Problem 1.65P
The Lewis structure of the given molecule is,
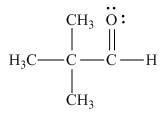
Figure 5
Explanation of Solution
Lewis structures can be drawn by following the below rules.
• Draw a carbon back bone of the compound.
• Each carbon atom has enough hydrogens to make it tetravalent.
• All heteroatoms and the hydrogens directly bonded to them are drawn.
To interpret a condensed formula one should start from left and proceed to right. All the carbon atoms are tetravalent. A carbon bonded to three

Figure 5
The Lewis structure of the given molecule is shown in Figure 5.
(f)
Interpretation: Condensed formula of the given molecule is to be converted to Lewis structure.
Concept introduction: Lewis structures can be drawn for organic compounds which contains both rings and chains of atoms. In Lewis structure compounds are represented with symbols of atoms connected with bonds.
Answer to Problem 1.65P
The the Lewis structure of the given molecule is,
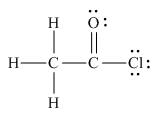
Figure 6
Explanation of Solution
Lewis structures can be drawn by following the below rules.
• Draw a carbon back bone of the compound.
• Each carbon atom has enough hydrogens to make it tetravalent.
• All heteroatoms and the hydrogens directly bonded to them are drawn.
To interpret a condensed formula one should start from left and proceed to right. All the carbon atoms are tetravalent. A carbon bonded to three
oxygen and chlorine is bonded to carbon atom. Thus, the Lewis structure of the given molecule is,

Figure 6
The Lewis structure of the given molecule is shown in Figure 6.
(g)
Interpretation: Condensed formula of the given molecule is to be converted to Lewis structure.
Concept introduction: Lewis structures can be drawn for organic compounds which contains both rings and chains of atoms. In Lewis structure compounds are represented with symbols of atoms connected with bonds.
Answer to Problem 1.65P
The Lewis structure of the given molecule is,
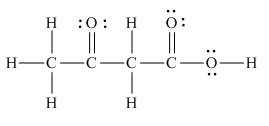
Figure 7
Explanation of Solution
Lewis structures can be drawn by following the below rules.
• Draw a carbon back bone of the compound.
• Each carbon atom has enough hydrogens to make it tetravalent.
• All heteroatoms and the hydrogens directly bonded to them are drawn.
To interpret a condensed formula one should start from left and proceed to right. All the carbon atoms are tetravalent. A carbon bonded to three

Figure 7
The Lewis structure of the given molecule is shown in Figure 7.
(h)
Interpretation: Condensed formula of the given molecule is to be converted to Lewis structure.
Concept introduction: Lewis structures can be drawn for organic compounds which contains both rings and chains of atoms. In Lewis structure compounds are represented with symbols of atoms connected with bonds.
Answer to Problem 1.65P
The Lewis structure of the given molecule is,
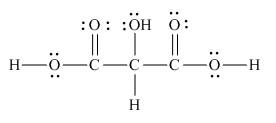
Figure 8
Explanation of Solution
Lewis structures can be drawn by following the below rules.
• Draw a carbon back bone of the compound.
• Each carbon atom has enough hydrogens to make it tetravalent.
• All heteroatoms and the hydrogens directly bonded to them are drawn.
To interpret a condensed formula one should start from left and proceed to right. All the carbon atoms are tetravalent. A carbon bonded to three

Figure 8
The Lewis structure of the given molecule is shown in Figure 8.
Want to see more full solutions like this?
Chapter 1 Solutions
Organic Chemistry-Package(Custom)
- Given the bonds C N, C H, C Br, and S O, (a) which atom in each is the more electronegative? (b) which of these bonds is the most polar?arrow_forwardDraw the Lewis structure (with formal charges) of the chemical species: 1. CINO2 (N is the central atom) 2. CO3^2- 3. NO2- 4. SO3 5. BrNO2 (N is the central atom) 6. HCO3- (H is bonded to one of the O's) 7. SeO2 8. HC2O4- (each C has two O atoms and H is on one of the O's) 9. HNO3 (H is bonded to one of the O's) 10. CH3NO2 11. HCO2- (H and both O's are bonded to C)arrow_forwardWrite the Lewis structure for each molecule.a. CH2O b. C2Cl4c. CH3NH2 d. CFCl3 (C central)arrow_forward
- Write the Lewis structure for each molecule or ion.a. H3COH b. OH- c. BrO- d. O22 -arrow_forwardWrite the Lewis structures for the following molecules, including all resonance forms, and show formal charges. The relative positions of the atoms are given as reference. 1. НСО2 H C H 2. CH2NO2 C N H H 3. CH2N2 C N N H.arrow_forward45. The Lewis structures for CH4, N2, and CO2, along with the corresponding space-filling molecular images, are shown here: Use Lewis theory and VSEPR theory to draw similar H space-filling molecular images of H-C-H a. CO b. NH3 :N=N: с. Н2S d. SİH4 :ö=c=ö:arrow_forward
- 1-What is the formal charge of the nitrogen atom in hydrogen cyanide (HCN)? a.5 b.−1 c.0 d.1 e.2 2-Which of the following diatomic molecules has the least number of valence electrons? a.N2 b.F2 c.O2 d.Cl2 e.NOarrow_forwardDraw the Lewis structure for each organic compound from its condensed structural formula.a. C2H4 b. CH3NH2 c. HCHOd. CH3CH2OH e. HCOOHarrow_forwardWrite the Lewis structure for each ion. Include resonance structures if necessary and assign formal charges to all atoms. If necessary, expand the octet on the central atom to lower formal charge.a. PO43- b. CN- c. SO3 2- d. ClO2 -arrow_forward
- Choose the best Lewis structure for CH2C12. H. a. H. 6. H–Cl–C-Cl-H H-H-C-Cl-Cl: H. Cl=c=Cl: d. Harrow_forward2. NH₂ a. Lewis structure e. Electronic geometry_ d. Molecular shape_ Bond angle? b. Draw the different bonds and label their polarity.arrow_forwardA. The Lewis diagram for PO There are HO B. The Lewis diagram for SeoBr2 is: Foot 3- The electron-pair geometry around the P atom in PO 19 There are M 808 lone pair(s) around the central atom, so the geometry of PO,3 is CEN :0-Se-Br: Br: "D The electron-pair geometry around the Se alom in SeoBr₂ is lone pair(s) around the central atom, so the geometry of SeoBr₂ isarrow_forward
 Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
