
Concept explainers
(a)
Interpretation:
To draw a valid Lewis structure for the given compound
Concept introduction:
In chemistry Lewis structure is very important as shown below in the Fig.1
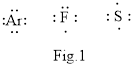
The above shown atoms combine to form molecule to attain stale noble gas configuration.
The structure in which all the electrons in the valence shells of the combining atoms are shown in the form of dots is called Lewis structure. The valence electrons that take in the bonding are called the bonding electrons and the valence electrons that are not participate is the nonbonding electrons.
(b)
Interpretation:
To draw a valid Lewis structure for the given compound
Concept introduction:
In chemistry Lewis structure is very important as shown below in the Fig.1
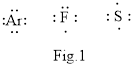
The above shown atoms combine to form molecule to attain stale noble gas configuration.
The structure in which all the electrons in the valence shells of the combining atoms are shown in the form of dots is called Lewis structure. The valence electrons that take in the bonding are called the bonding electrons and the valence electrons that are not participate is the nonbonding electrons.
(C)
Interpretation:
To draw a valid Lewis structure for the given compound
Concept introduction:
In chemistry Lewis structure is very important as shown below in the Fig.1
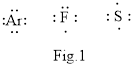
The above shown atoms combine to form molecule to attain stale noble gas configuration.
The structure in which all the electrons in the valence shells of the combining atoms are shown in the form of dots is called Lewis structure. The valence electrons that take in the bonding are called the bonding electrons and the valence electrons that are not participate is the nonbonding electrons.
(d)
Interpretation:
To draw a valid Lewis structure for the given compound
Concept introduction:
In chemistry Lewis structure is very important as shown below in the Fig.1
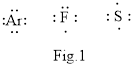
The above shown atoms combine to form molecule to attain stale noble gas configuration.
The structure in which all the electrons in the valence shells of the combining atoms are shown in the form of dots is called Lewis structure. The valence electrons that take in the bonding are called the bonding electrons and the valence electrons that are not participate is the nonbonding electrons.
(e)
Interpretation:
To draw a valid Lewis structure for the given compound
Concept introduction:
In chemistry Lewis structure is very important as shown below in the Fig.1
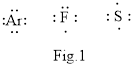
The above shown atoms combine to form molecule to attain stale noble gas configuration.
The structure in which all the electrons in the valence shells of the combining atoms are shown in the form of dots is called Lewis structure. The valence electrons that take in the bonding are called the bonding electrons and the valence electrons that are not participate is the nonbonding electrons.
Want to see the full answer?
Check out a sample textbook solution
Chapter 1 Solutions
Organic Chemistry-Package(Custom)
- 1.44 Draw one valid Lewis structure for each compound. Assume the atoms are arranged as drawn. H HCC NO H a. CH₂N₂ HCNN H H b. CH₂NO₂ HCNO HO C. CH3CNO d. (CH₂CN) HC CN H 1.45 Draw an acceptable Lewis structure from each condensed structure, such that all atoms have zero formal charge. a. diethyl ether, (CH3CH₂)2O, the first general anesthetic used in medical procedures b. acrylonitrile, CH₂CHCN, starting material used to manufacture synthetic Orlon fibers c. dihydroxyacetone, (HOCH₂)₂CO, an ingredient in sunless tanning products d. acetic anhydride, (CH3CO)2O, a reagent used to synthesize aspirinarrow_forwardd. H E Assign formal charges to each carbon atom in the given species. All lone pairs have been drawn in. H H a. CH2-CH b. H-C-H c. H-C-H ннarrow_forwardDraw a valid Lewis structure for each species.a. CH3CH3b. CH5Nc. CH3Clarrow_forward
- Draw the Lewis structure for each organic compound from its condensed structural formula.a. C2H4 b. CH3NH2 c. HCHOd. CH3CH2OH e. HCOOHarrow_forwardents V Soul (TV series) O REPRESENTATIONS OF ORGANIC MOLECULES Drawing a skeletal structure from a Lewis structure Convert the Lewis structure below into a skeletal structure. Н—С— Н H. H- H- Н—С—Н H. HH H. H- Н—С—С—С—С—С—С—С- Н C=C H- H. H. H. Н—С— Н H. ct+ Click and drag to start drawing a structure. HIC CIH HICarrow_forwardb. There is one additional resonance structure. NH₂ c. There is a total of five resonance structures (including the original structure). : OHarrow_forward
- Convert each condensed formula to a Lewis structure. a. CH3(CH2)4CH(CH3)2 b. (CH3)3CCH(OH)CH2CH3 c. (CH3)2CHCHO d. (HOCH2)2CH(CH2)3C(CH3)2CH2CH3arrow_forward1. Convert the condensed formulas to Lewis structures (show all atoms, bonds, lone pairs) a. CH3(CH₂)4OH b. CH3(CH₂)4CH(CH3)2 C. (CH3)2CHO(CH2)4CH3 d. (CH3)2CHCHOarrow_forwardDraw the Lewis structure for each organic compound from its condensed structural formula.a. C3H8 b. CH3OCH3 c. CH3COCH3d. CH3COOH e. CH3CHOarrow_forward
- 1. What is the relationship between the following compounds? H :0: H H:0: H H-C=C-C-C-C-H H-C-C-C=Ċ-H H HH H. H-C-H B. constitutional isomers C. the same structure A. isotopes D. composed of different elements E. no relationship 2. What is the correct Lewis structure for HN3, including the formal charges? CIHarrow_forwardThe Structure of Hydrocarbons 1. Draw the four carbon bonding patterns commonly seen in hydrocarbons and all other organic compounds. Construct the Lewis structures of the following compounds by combining the carbon atoms with the appropriate bonding patterns and the required number of hydrogen atoms. 2. a. hexane: CH,CH,CH,CH,CH,CH, b. 3-hexene: CH,CH,CH=CHCH,CH, c. 2-hexyne: CH,C=CCH,CH,CH, 3. There are three distinct bond angles formed in the four bonding patterns of carbon. What are they and in which bonding pattern(s) is each seen?arrow_forwardAssign formal charges to each carbon atom in the given species. All lone pairs have been drawn in. нн a. CH2=CH b. H-с-н с. Н-с-н d. H-С-С H H e. -N: f. g. h. :N=N=N: -N=O:arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





