
Interpretation:The missing values in the table are to be filled.
Concept introduction: The reagent in reaction that controls the amount of product formed is termed as limiting reagent. After completion of
Answer to Problem 4E
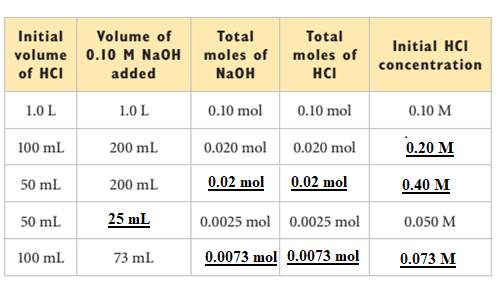
The missing values in the table are filled as shown below:
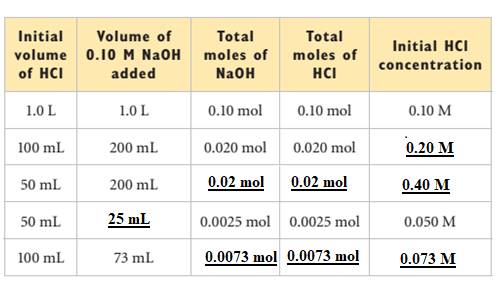
Explanation of Solution
The reaction between
Here, the
From the II data, the initial concentration of HCl is calculated as shown below:
From the III data, the number of moles of NaOH,
The number of moles of HCl,
The initial concentration of HCl,
From the IV data, the volume of NaOH,
From the V data, the number of moles of NaOH,
The number of moles of HCl,
The initial concentration of HCl,
Therefore, the given is filled as shown below:
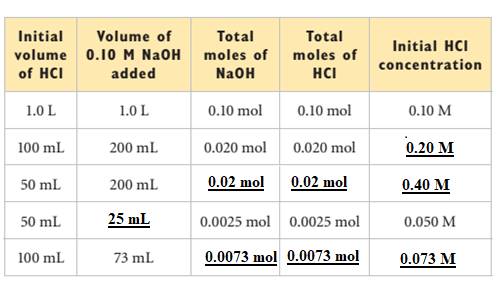
The missing values in the table are filled as shown below:
Chapter U4 Solutions
Living by Chemistry
Additional Science Textbook Solutions
Chemistry: Structure and Properties
Chemistry: The Central Science (14th Edition)
Organic Chemistry
Introductory Chemistry (5th Edition) (Standalone Book)
Chemistry: A Molecular Approach (4th Edition)
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





