
Study Guide for Campbell Biology
11th Edition
ISBN: 9780134443775
Author: Lisa A. Urry, Michael L. Cain, Steven A. Wasserman, Peter V. Minorsky, Jane B. Reece, Martha R. Taylor, Michael A. Pollock
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 8, Problem 7IQ
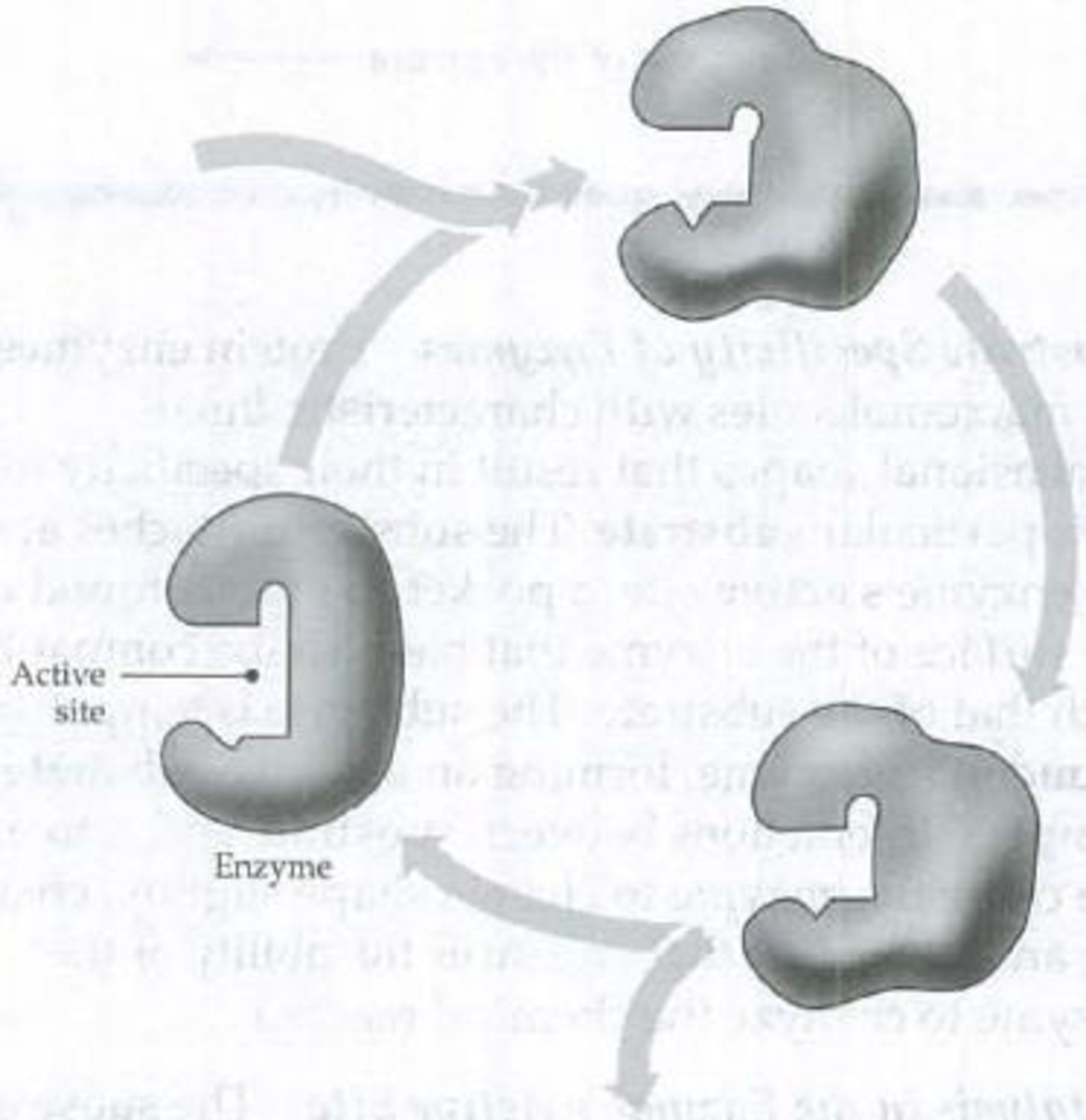
In the following diagram of a catalytic cycle, sketch two appropriate substrate molecules and two products, show the substrate and product molecules in the active site, and label the key steps of the cycle.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Briefly describe 3 general catalytic mechanisms and 2 catalytic mechanisms of enzymes
Which of the following describe what enzymes such as Chymotrysin accomplish to allow products to form and be released by the enzyme active site?
Group of answer choices
catalyze chemical eactions that are thermodynamically unfavorable to occur
establish a more stable transition state at a higher energy level to perform a catalysis reaction
redce the chemical energy held within a chemical bond
modify chemical structure of a substrate to "fit" within the active site
Which of the following statements is true for the shown reaction?
Substrate level phosphorylation occurs during the reaction
Phosphoryl transfer occurs during the reaction
Both A and B
Neither A nor B
Chapter 8 Solutions
Study Guide for Campbell Biology
Ch. 8 - Complete the following concept map that summarizes...Ch. 8 - Complete the following table to indicate how the...Ch. 8 - Develop a concept map on free energy and G. The...Ch. 8 - Prob. 4IQCh. 8 - Prob. 5IQCh. 8 - In the following graph of an exergonic reaction...Ch. 8 - In the following diagram of a catalytic cycle,...Ch. 8 - Return to the diagram in Interactive Question 3.7,...Ch. 8 - Both ATP and ADP serve as regulators of enzyme...Ch. 8 - What is the relationship between the concept of...
Ch. 8 - What role do enzymes play in metabolism?Ch. 8 - ________ the totality of an organisms chemical...Ch. 8 - _______ pathways that use energy to synthesize...Ch. 8 - Prob. 3TYKFCh. 8 - _______ the most random form of energyCh. 8 - _______ term for the measure of disorder or...Ch. 8 - Prob. 6TYKFCh. 8 - _______ inhibitors that decrease an enzymes...Ch. 8 - Prob. 8TYKFCh. 8 - Prob. 9TYKFCh. 8 - Prob. 10TYKFCh. 8 - Catabolic and anabolic pathways are often coupled...Ch. 8 - Which statement most closely reflects the first...Ch. 8 - When a cell breaks down glucose, only about 34% of...Ch. 8 - Prob. 4TYKCh. 8 - Prob. 5TYKCh. 8 - Prob. 6TYKCh. 8 - One way in which a cell maintains metabolic...Ch. 8 - Prob. 8TYKCh. 8 - Prob. 9TYKCh. 8 - What is meant by an induced fit? a. The binding of...Ch. 8 - In an experiment, changing the pH from 7 to 6...Ch. 8 - Prob. 12TYKCh. 8 - Penicillin binds to the active site of an enzyme...Ch. 8 - Prob. 14TYKCh. 8 - Prob. 15TYKCh. 8 - Which line in the diagram indicates the G of the...Ch. 8 - Prob. 17TYKCh. 8 - Prob. 18TYKCh. 8 - Prob. 19TYKCh. 8 - Prob. 20TYK
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biology and related others by exploring similar questions and additional content below.Similar questions
- Inhibitors are compounds capable of blocking the catalytic process. Outline with the use of graphs and equations in illustrating the different modes of action of enzyme inhibitors.arrow_forwardWhich of the following statements regarding enzyme catalysis is false? All options are false. Once formed, the transition state slowly proceeds to forming the product at a rate determined by cofactor binding The free energy of binding of the enzyme to the transition state is more favorable than the free energy of binding of the enzyme to the substrate The substrate and active site of the enzyme are solvated to promote enzyme-substrate interaction Once formed, the product dissociates from the enzyme after ATP hydrolysis in order to regenerate the active sitearrow_forwardComplete the following diagram, using arrows to show the flow of electrons, for this reaction catalyzed by GAP dehydrogenase. Draw how the enzyme pocket appears as the reaction is completed. Indicate the product (if any). NAD+ ÇHOPO он Сysarrow_forward
- Briefly describe 3 general catalytic mechanismsarrow_forwardWhich of the following statements is true for the shown reaction? The reaction can occur in both cytosol and mitochondria Under starved conditions, the reaction becomes reversible to allow the synthesis of ketonebodies The reaction requires FMN as a cofactor Increase in NADH stimulates the reaction None of the abovearrow_forwardAt what temperature in the above graph is the enzyme the most active?arrow_forward
- Consider the following free energy diagram for an uncatalyzed and enzyme-catalyzed reaction. Select all the statements that are true. Without enzyme With enzyme A+B Time AB Oa. The reaction is now spontaneous due to the addition of enzyme b. The rate of the enzyme catalyzed reaction is faster than the uncatalyzed reaction O C. The reaction is exergonic O d. The change in free energy for the reaction is greater in the catalyzed reaction, compared to the uncatalyzed reaction e. The enzyme stabilizes the transition state for the reaction Released Energy pesarrow_forwardlist three factor which influence enzymatic activity and list the four steps of cellular respiration and where they take place in the cell. Write the equation for cellular reespiration and photosynthesis indicate which reactant is oxdidized and which is reduced in both equationarrow_forwardThe figure below represents a reaction in the presence and absence of an enzyme catalyst. What can you conclude about this reaction? energy activation energy activation energy 1 uncatalysed reaction catalysed reaction A) It is endergonic in the absence of the enzyme. B) It will proceed more quickly in the presence of the enzyme. C) Once the reaction has reached equilibrium, there will be more product in the enzyme-catalyzed reaction. D) It has a negative AG. OE) Both B and D are truearrow_forward
- An allosteric enzyme that follows the concerted mechanism has a T/R ratio of 500 in the absence of substrate. Suppose that a mutation reversed the ratio. How would this mutation affect the relation between the rate of the reaction and substrate concentration? The mutant enzyme would behave like an enzyme that obeys Michaelis Menton kinetics. The mutant enzyme would have a smaller vmax There would be no difference in the mutant enzyme in terms of substrate binding and catalysis. More than one answer is correct. The mutant enzyme would display cooperativity more than the wild type. MacBook Air 888 F5 F4 F3 F2 %23 2$ %arrow_forwardWhich of the following is true under the following conditions: an enzyme displaying Michaelis-Menten kinetics where the enzyme concentration is 10 nM, the substrate concentration is 45 mM, and the Km is 50 µM? a) The enzyme has low catalytic efficiency for the substrate. b)The rate of catalysis is near half-maximal velocity. c)The enzymatic reaction is near maximal velocity. d)Halving the substrate concentration has little effect on the catalytic rate. e) There is not enough information provided.arrow_forwardThe above reaction coordinate diagram in the presence of enzyme is inconsistent with the "lock and key" model of enzyme-substrate l binding. Draw and properly label the reaction. coordinate diagram for the enzymatic reaction according to the "lock and key" model. Explain why this model is inconsistent with catalytic properties of enzymes.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON
Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax
Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education,
Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education, Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company
Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co.
Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co. Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education
Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education

Human Anatomy & Physiology (11th Edition)
Biology
ISBN:9780134580999
Author:Elaine N. Marieb, Katja N. Hoehn
Publisher:PEARSON

Biology 2e
Biology
ISBN:9781947172517
Author:Matthew Douglas, Jung Choi, Mary Ann Clark
Publisher:OpenStax

Anatomy & Physiology
Biology
ISBN:9781259398629
Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa Stouter
Publisher:Mcgraw Hill Education,

Molecular Biology of the Cell (Sixth Edition)
Biology
ISBN:9780815344322
Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter Walter
Publisher:W. W. Norton & Company

Laboratory Manual For Human Anatomy & Physiology
Biology
ISBN:9781260159363
Author:Martin, Terry R., Prentice-craver, Cynthia
Publisher:McGraw-Hill Publishing Co.

Inquiry Into Life (16th Edition)
Biology
ISBN:9781260231700
Author:Sylvia S. Mader, Michael Windelspecht
Publisher:McGraw Hill Education
The Cell Membrane; Author: The Organic Chemistry Tutor;https://www.youtube.com/watch?v=AsffT7XIXbA;License: Standard youtube license