
Study Guide for Campbell Biology
11th Edition
ISBN: 9780134443775
Author: Lisa A. Urry, Michael L. Cain, Steven A. Wasserman, Peter V. Minorsky, Jane B. Reece, Martha R. Taylor, Michael A. Pollock
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 8, Problem 16TYK
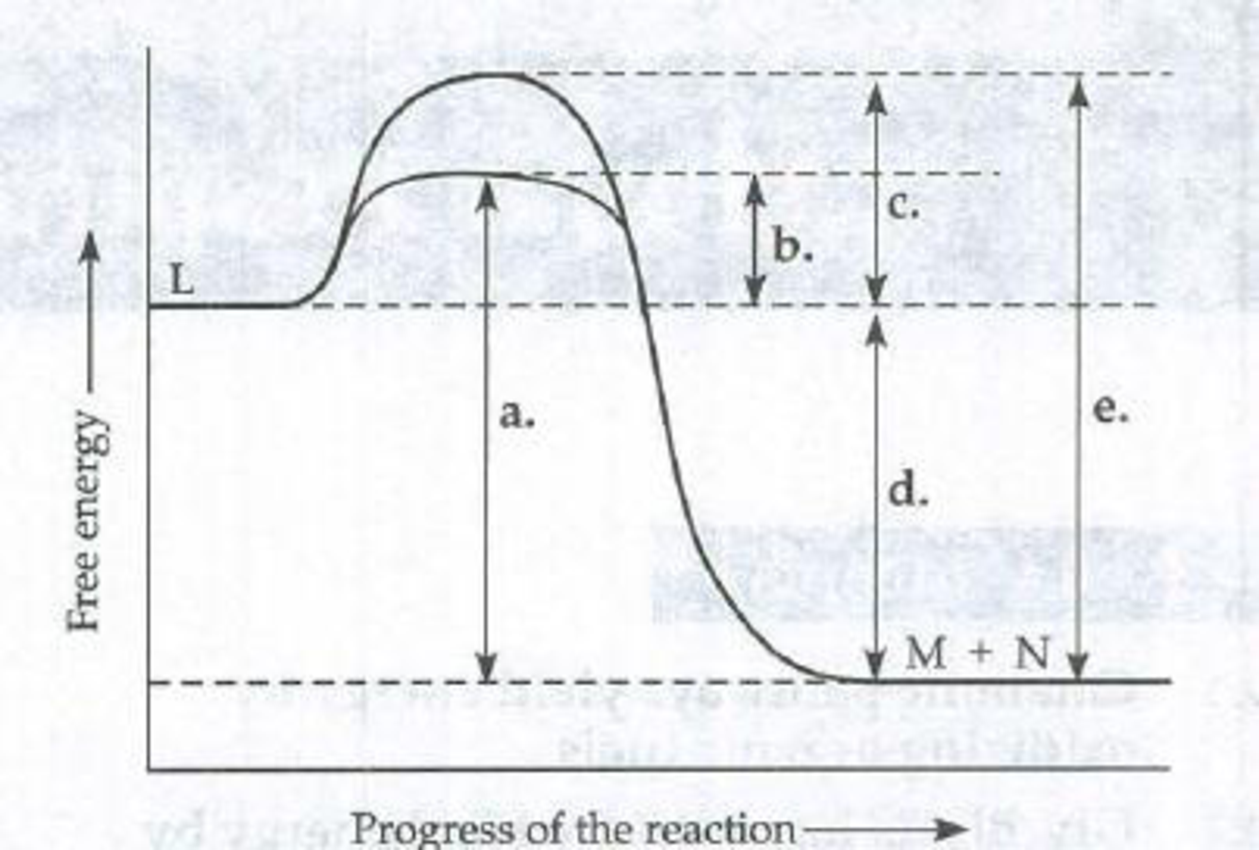
Which line in the diagram indicates the ΔG of the enzyme-catalyzed reaction L → M + N?
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Given the following enzyme-catalyzed reaction, identify the class and subclass of the enzyme involved.
HO
Class: [Select]
COO™
NH3*
Subclass: [Select]
H₂O
H₂C=
COO™
NH3*
Given the following enzyme-catalyzed reaction, identify the class and subclass of the enzyme involved.
H
H
e
ООС
Class: [Select]
Subclass: [Select]
COO
-H
H
+ FAD
H
ООС
COO
+ FADH₂
Which type of enzyme (Table) catalyzes the following (given) reactions?
Chapter 8 Solutions
Study Guide for Campbell Biology
Ch. 8 - Complete the following concept map that summarizes...Ch. 8 - Complete the following table to indicate how the...Ch. 8 - Develop a concept map on free energy and G. The...Ch. 8 - Prob. 4IQCh. 8 - Prob. 5IQCh. 8 - In the following graph of an exergonic reaction...Ch. 8 - In the following diagram of a catalytic cycle,...Ch. 8 - Return to the diagram in Interactive Question 3.7,...Ch. 8 - Both ATP and ADP serve as regulators of enzyme...Ch. 8 - What is the relationship between the concept of...
Ch. 8 - What role do enzymes play in metabolism?Ch. 8 - ________ the totality of an organisms chemical...Ch. 8 - _______ pathways that use energy to synthesize...Ch. 8 - Prob. 3TYKFCh. 8 - _______ the most random form of energyCh. 8 - _______ term for the measure of disorder or...Ch. 8 - Prob. 6TYKFCh. 8 - _______ inhibitors that decrease an enzymes...Ch. 8 - Prob. 8TYKFCh. 8 - Prob. 9TYKFCh. 8 - Prob. 10TYKFCh. 8 - Catabolic and anabolic pathways are often coupled...Ch. 8 - Which statement most closely reflects the first...Ch. 8 - When a cell breaks down glucose, only about 34% of...Ch. 8 - Prob. 4TYKCh. 8 - Prob. 5TYKCh. 8 - Prob. 6TYKCh. 8 - One way in which a cell maintains metabolic...Ch. 8 - Prob. 8TYKCh. 8 - Prob. 9TYKCh. 8 - What is meant by an induced fit? a. The binding of...Ch. 8 - In an experiment, changing the pH from 7 to 6...Ch. 8 - Prob. 12TYKCh. 8 - Penicillin binds to the active site of an enzyme...Ch. 8 - Prob. 14TYKCh. 8 - Prob. 15TYKCh. 8 - Which line in the diagram indicates the G of the...Ch. 8 - Prob. 17TYKCh. 8 - Prob. 18TYKCh. 8 - Prob. 19TYKCh. 8 - Prob. 20TYK
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biology and related others by exploring similar questions and additional content below.Similar questions
- In a typical enzyme-catalyzed reaction, what are the relative concentrations of reactants and products as compared to the enzyme concentration?arrow_forwardIn the scheme below which represents the mechanism of action for a large number of enzymes: A+B⟺AB⟶C The steady state approximation is reached when: d[AB]/dt≈0 k2≫k1 k−1≫k1 k−1=k1arrow_forwardConsider the following free energy diagram for an uncatalyzed and enzyme-catalyzed reaction. Select all the statements that are true. Without enzyme With enzyme A+B Time AB O a. The rate of the enzyme catalyzed reaction is faster than the uncatalyzed reaction O b. The change in free energy for the reaction is greater in the catalyzed reaction, compared to the uncatalyzed reaction O c. The enzyme stabilizes the transition state for the reaction Od. The reaction is exergonic е. The reaction is now spontaneous due to the addition of enzyme Released Energyarrow_forward
- What are the rate constants for the enzyme-catalyzed reaction?arrow_forwardConsider an enzyme that catalyzes the reaction S2 P, by the following simple reaction mechanism: k, E + S 2 E•S →E kcat + P Suppose the enzyme acquires a mutation that causes k1 to be 10-times smaller than for the wild-type (non-mutant) enzyme. Suppose you measure the initial reaction rate (vo) at several different [S] for the mutant and the wild-type enzymes. Under what conditions would the mutation have a greater effect on the reaction rate (vo) of the mutant enzyme compared to the wild-type enzyme - at very low [S], or at very high [S]? Explain briefly how you decided.arrow_forwardSketch the complete reaction free energy diagram for an enzyme-catalyzed conversion of a single substrate (S) into product (P), where the reaction is spontaneous in the forward direction Overlay the free energy diagram for the uncatalyzed reaction and indicate delta delta G〒 on your sketch: Chemical step is rate limitingarrow_forward
- The enzyme phosphoglucomutase catalyzes the conversion of glucose 1-phosphate to glucose 6-phosphate. After the reactants and products were mixed and allowed to reach equilibrium at 25°C, the concentration of glucose 1-phosphate was 4.5 mM and that of glucose 6-phosphate was 86 mM. Calculate Keq' and AG for this reaction. The reaction coordinate diagram for an enzyme-catalyzed reaction is shown below. How many transition states and intermediates are in the reaction? Is the reaction thermodynamically favorable? Which step is the rate-determining step of the reaction? G Reaction coordinatearrow_forwardWhat class of enzyme catalyzes the following reaction?arrow_forwardUse the data below to determine the maximum velocity [in mM/s] of a certain enzyme-catalyzed reaction. v = 0.152 mM/s at [S] = 0.334 mM v = 0.190 mM/s at [S] = 0.450 mMarrow_forward
- Given the following data in enzyme-catalyzed reaction, what are the Vm, Km of with DEDS (presence of inhibitor) and without DEDS ( absence of inhibitor) and its type of inhibition.arrow_forwardWhat general effects would you expect the following changes to have on the rate of an enzyme-catalyzed reaction for an enzyme that has its maximum activity at body temperature (about 37 °C/310.15 K)?(a) Lowering the reaction temperature from 313 K (40 °C)to 283 K (10 °C)(b) Adding a drop of a dilute HgCl2 solution(c) Adding an oxidizing agent, such as hydrogen peroxidearrow_forwardWhat does it mean to say that an enzyme- catalyzed reaction is either enzyme limited or substrate -limited?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON
Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax
Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education,
Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education, Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company
Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co.
Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co. Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education
Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education

Human Anatomy & Physiology (11th Edition)
Biology
ISBN:9780134580999
Author:Elaine N. Marieb, Katja N. Hoehn
Publisher:PEARSON

Biology 2e
Biology
ISBN:9781947172517
Author:Matthew Douglas, Jung Choi, Mary Ann Clark
Publisher:OpenStax

Anatomy & Physiology
Biology
ISBN:9781259398629
Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa Stouter
Publisher:Mcgraw Hill Education,

Molecular Biology of the Cell (Sixth Edition)
Biology
ISBN:9780815344322
Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter Walter
Publisher:W. W. Norton & Company

Laboratory Manual For Human Anatomy & Physiology
Biology
ISBN:9781260159363
Author:Martin, Terry R., Prentice-craver, Cynthia
Publisher:McGraw-Hill Publishing Co.

Inquiry Into Life (16th Edition)
Biology
ISBN:9781260231700
Author:Sylvia S. Mader, Michael Windelspecht
Publisher:McGraw Hill Education
Enzyme Kinetics; Author: MIT OpenCourseWare;https://www.youtube.com/watch?v=FXWZr3mscUo;License: Standard Youtube License