
Interpretation:
Model depicting the property of malleability and ductility based on sea of electron needs to be drawn.
Concept introduction:
Metallic solid- Solids which are composed of metal atoms that are held by metallic bonds wherein the electrons are delocalized and metal ion is embedded in sea of electrons is a metallic solid. It is like a huge molecular orbital that spans across the whole solid
Answer to Problem 45SSC
The model that depicts the property of malleability and ductility is having metal ions surrounded on all sides by electrons.
Explanation of Solution
The solids that are composed of metal atoms which are held together by metallic bonds are termed as metallic solids. It is considered to be a big molecular orbital which spans the whole of the solid. This indicates that electrons in the metallic solids are delocalized.
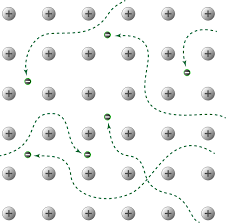
Due to the spread of electrons among the lattice of positively charged ions, the electrons spread its density over all atoms. When it is beaten, it can be observed that the layers of atoms can roll over each other into new positions but without breaking metallic bonds. In other words, when it is under stress, the layers of atoms will roll over each other and on release of stress, it will come back to its original position.
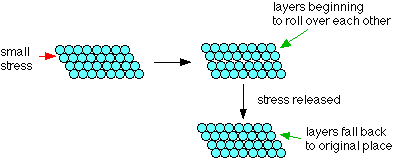
The model that depicts the property of malleability and ductility is having metal ions surrounded on all sides by electrons.
Chapter 7 Solutions
Glencoe Chemistry: Matter and Change, Student Edition
Additional Science Textbook Solutions
Organic Chemistry
CHEMISTRY-TEXT
Chemistry: Structure and Properties
Essential Organic Chemistry (3rd Edition)
Introductory Chemistry (6th Edition)
Chemistry: The Central Science (14th Edition)
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





