
Interpretation:
The formation of ionic compound from elements Strontium and fluorine needs to be explained.
Concept introduction:
Chemical compounds are formed when the atoms, ions or molecules of two substances are attracted towards each other forming a
Answer to Problem 9PP
1 Strontium will lose 2 electrons which is gained by 2 of Fluorine atoms to form ionic compound,
Explanation of Solution
The formation of Strontium Fluoride is when Strontium atoms interact with fluorine atom.
The electronic configuration of Strontium is
Strontium atom has two outermost electron which can be lost to become a positive ion.
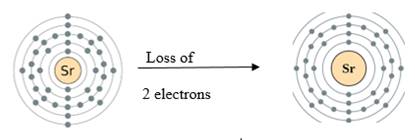
Figure 1
The electronic configuration of fluorine is
Fluorine atom has 1 half filled shells of p orbital. To fill up the half filled shell, it can take up 1 electron to become a fluoride anion.
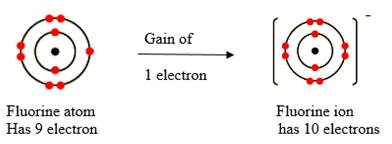
Figure 2
The 2 electrons lost by Strontium can be taken up by 2 of Fluorine atoms and form ionic compound. The Strontium is positively charged and Fluorine ion is negatively charged which gets attracted to form ionic compound.
Hence, we can say that one Strontium atom bonds with 2 Fluorine atoms to form ionic compound. The 2 electrons of Strontium gets gained by Fluorine to get an octet structure and form ionic compound,
1 Strontium will lose 2 electrons which is gained by 2 of Fluorine atoms to form ionic compound
Chapter 7 Solutions
Glencoe Chemistry: Matter and Change, Student Edition
Additional Science Textbook Solutions
Microbiology with Diseases by Body System (5th Edition)
College Physics: A Strategic Approach (3rd Edition)
Introductory Chemistry (6th Edition)
Chemistry: The Central Science (14th Edition)
Campbell Biology: Concepts & Connections (9th Edition)
Human Physiology: An Integrated Approach (8th Edition)
- Indicate whether in laminar materialsa) all the sheets are neutral.b) all the sheets have a negative charge.c) all the sheets have a positive charge.d) all of the above are correct.arrow_forwardPropose a mechanism for reaction of the first three propylene units in the polymerization of propylene in the presence of a peroxide. n H₂C=CHCH3 propylene ROOR high pressure H H C-C H CH3 n polypropylenearrow_forwardNonearrow_forward
- For the E2 reaction below, predict the major organic products. If no elimination products are formed, check the box below the drawing area. Br + © Na OH Click and drag to start drawing a structure.arrow_forwardPlease don't use Ai solutionarrow_forwardUse the periodic table below to answer the question that follows. The Periodic Table of Elements 2 1 Nonmetals Noble Gases H 1.008 Hydrogen 4 11 19 6.94 Lithium Na 22.990 Sodium Be 9.012 Beryllium 12 Mg 24.305 Magnesium 20 Ca 40.08 Calcium 21 Sc 44.956 Scandium Transition Metals Alkali Metals Alkaline Earth Metals Lanthanides Actinides 22 Ti 47.867 Titanium 23 V 50.942 Vanadium Post-Transition Metals Metalloids 40 Zr 91.224 Zirconium 88.906 Yttrium 57-71 72 K 39.098 Potassium 37 Rb 85.468 Rubidium 55 87 Cs 132.905 Cesium Fr 223.020 Francium 38 39 Sr 87.62 Strontium 56 88 Ba 137.328 Barium Ra 226.025 Radium Hf 178.49 Hafnium 89-103 104 Rf [261] Rutherfordium 41 Nb 92.906 Niobium 73 Ta 180.948 Tantalum 105 Db 24 Halogens Cr 51.996 Chromium 42 25 Mn 54.938 Manganese 43 TC Mo 95.95 Molybdenum Technetium 98.907 26 44 Fe 55.845 Iron Ru 101.07 Ruthenium 74 75 76 W Re 183.84 Tungsten 106 Sg 186.207 Rhenium 107 Bh 27 45 Co 58.933 Cobalt Rh 102.906 Rhodium 28 46 Ni 58.693 Nickel Pd 106.42…arrow_forward
- Use the periodic table below to answer the question that follows. The Periodic Table of Elements 1 5 13 B 10.811 Boron Al 26.982 Aluminum 31 49 Ga 69.723 Gallium 26 Fe 55.845 Iron Ru 101.07 Ruthenium Os 192.217 Osmium Hs 27 45 Co 58.933 Cobalt Rh 102.906 Rhodium 77 Ir 192.217 Iridium 109 Mt 28 46 Ni 58.693 Nickel Pd 106.42 Palladium 78 Pt 195.085 Platinum 110 29 47 79 30 Cu Zn 63.546 Copper Ag 107.868 Silver Au 196.967 Gold 48 65.38 Zinc Cd 112.411 Cadmium 80 Hg 81 114.818 Indium Tl 6 14 32 C 12.011 Carbon Si 28.086 Silicon Ge 72.631 Germanium 50 Sn 118.711 Tin 7 8 9 N FL 14.007 Nitrogen 15.999 Oxygen 18.998 Fluorine 16 17 15 30.974 Phosphorus 33 51 As 74.922 Arsenic Sb 121.760 Antimony Bi 34 32.066 Sulfur Se 78.972 Selenium 52 Te 127.6 Tellurium 84 Po [208.982] Polonium 35.453 Chlorine 35 53 85 Br 79.904 Bromine 126.904 lodine At 209.987 Astatine 10 18 He 4.003 Helium Ne 20.180 Neon 36 54 86 Ar 39.948 Argon Kr 84.798 Krypton Xe 131.294 Xenon Rn 222.018 82 83 Pb 200.592 Mercury 204.383…arrow_forwardm Given that the freezing point of water is 0.0 °C at 1 atm and the freezing point depression constant is 1.86, what is the freezing point of a 0.05 m solution of a nonvolatile nonelectrolyte solute in water? • Your answer should have one significant figure. Provide your answer below: degrees Carrow_forwardmol Malate dehydrogenase catalyzes the oxidation of malate to oxaloacetate with a standard Gibbs free energy of AG° = +29.7*. What is the equilibrium constant (Keq) for this reaction at 298 K and pH 7? Under physiological conditions at 310 K and pH 7, this reaction is at equilibrium. In the mitochondrial matrix, the ratio NAD+/NADH is 8 and the concentration of malate is 1.0 mM. What is the concentration of oxaloacetate? Malate dehydrogenase is also present in the cytoplasm where the ratio NAD+/NADH is 700 and malate concentration is 1.0 mM. If this reaction is at equilibrium at 310 K and pH 7, what is the cytosolic concentration of oxaloacetate? malate + NAD+ oxaloacetate + NADH + H+arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





