
Concept explainers
Interpretation:
The property of malleability and ductility needs to be explained based on metallic bonding.
Concept introduction:
The bonding between two or more atoms, molecules or ions results in
Answer to Problem 89A
The metallic bonds are those which have metal ions in sea of electrons which will move in direction of the force applied and hence said to be malleable and ductile.
Explanation of Solution
Metallic bonds are formed as a result of overlapping of electronic orbitals. The electrons present in the electronic orbitals are shared between the atoms. The vacant p and d orbitals in outer energy levels of a metal allows the free movement of electrons. In other words, this sea of electrons (negatively charged) attract positive kernels and is the main reason for conductivity, malleability and the metallic bonding in a metal.
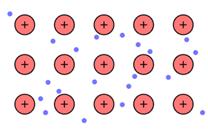
The positive ion (kernels) that flows in the negatively charged sea of electrons is the main reason of metallic bonding.
Hence, when metals are beaten these delocalized electrons moves in the direction of the force applied and therefore can be beaten to sheets or rolled into wires easily.
The property of positively charged ions to move from one place to another in the negatively charged sea makes the metal malleable and ductile. Each metal have a different tendency to be drawn into wires or beaten into sheets as per the movement of these kernels.
The metallic bonds are those which have metal ions in sea of electrons which will move in direction of the force applied and hence said to be malleable and ductile.
Chapter 7 Solutions
Glencoe Chemistry: Matter and Change, Student Edition
Additional Science Textbook Solutions
Chemistry: Structure and Properties
CHEMISTRY-TEXT
Chemistry: A Molecular Approach (4th Edition)
Chemistry: Structure and Properties (2nd Edition)
General Chemistry: Principles and Modern Applications (11th Edition)
Chemistry: The Central Science (13th Edition)
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





