
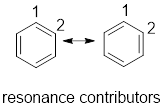
Concept explainers
(a)
Interpretation:
From the given compounds, compounds having delocalised electrons have to be identified.
Concept Introduction:
Delocalized electrons: Electrons that are not localized in one particular atom or bond and shared by three or more atoms are called delocalized electrons.
Resonance contributor: In some chemical compounds like benzene pi electrons are delocalized in it and difficult to locate it. Resonance contributor gives an idea of where about of pi electrons. The exact structure with localized electrons are called resonance contributor.
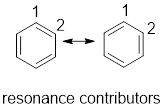
(a)
Interpretation:
From the given compounds, resonance contributor for compounds having delocalised electrons have to be drawn.
Concept Introduction:
Delocalized electrons: Electrons that are not localized in one particular atom or bond and shared by three or more atoms are called delocalized electrons.
Resonance contributor: In some chemical compounds like benzene pi electrons are delocalized in it and difficult to locate it. Resonance contributor gives an idea of whereabouts of pi electrons. The exact structure with localized electrons are called resonance contributor.
Want to see the full answer?
Check out a sample textbook solution
Chapter 7 Solutions
Essential Organic Chemistry, Global Edition
- H. H H. H-C-0-C* H---0=C Н—С— H. H. H. H. Which of the following resonance structures is the more major contributor, the one on the left or the one on the right? left O rightarrow_forward7. Draw all A. B. Ho important resonance structures for each:arrow_forwardRank the resonance structures in each group in order of increasing contribution to the resonance hybrid. Label the resonance structure that contributes the most as 3 and the resonance structure that contributes the least as 1. Label the intermediate contributor as 2. :O: a. CH3-C-ö-cCH, CH3-C-ö-CH3 CH,-C=0-CH, ÇH3 CH3-C-N=NH2 CH3 ÇH3arrow_forward
- Complete the fourth resonance structure to determine which bond is shortest? Which compound will react fourth fastest with B-eBr ? d. e. F 14. Complete the fourth resonance structure to determine which bond is shortest? 15. Which compound will react fourth fastest with Br₂/FeBr3? a. b. d. -OH b darrow_forwardLabel each double bond E, Z, or neither. (It may help to draw in some missing H’s.)arrow_forwardFor each proposed set of resonance structures: a. (E) Add curved arrows (starting from left) to show how each successive r.s. was generated. b. Cross out any resonance structures that are NOT important, and explain your reasoning.arrow_forward
- a. What is the hybridization of each N atom in nicotine? b. What is the geometry around each N atom? c. In what type of orbital does the lone pair on each N atom reside? d. Draw a constitutional isomer of nicotine. e. Draw a resonance structure of nicotine.arrow_forward4. Draw the possible resonance structures for compounds C and D. Include lone pairs and indicate non-zero formal charges. Circle the minor resonance contributor for compound C. O :O: :O: C & Darrow_forward4. The purine heterocycle occurs commonly in the structure of DNA. a. How is each N atom hybridized? b. In what type of orbital does each lone pair on a N atom reside? c. How manyn electrons does purine contain? d. Why is purine aromatic? :N H. purine batond ai enexerlo 08,H HO HO HO b w bemmot etbubong erit wea OH8 HOH2O zhemala eriT nertw bemotarrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
