
Interpretation:
The compounds having delocalized electrons have to be predicted.
Concept Introduction:
Resonance Contributor: The appropriate structure with the delocalized electrons is called a resonance contributor, a resonance structure, or a contributing resonance structure.
Delocalized electrons: The sharing of electrons between two or more atoms known as delocalization of electrons. In order to have delocalized electrons, the system must be planar and have alternative double bonds and single bonds.
Resonance hybrid: The actual structure with delocalized electrons is called a resonance hybrid.
Explanation of Solution
Reason for correct options:
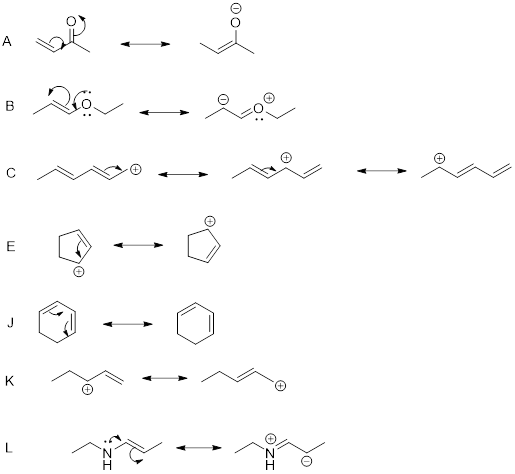
The above compounds all have delocalized electrons, which undergo electron movement.
The resonance structures can be drawn as shown above.
Hence, the correct options are A, B, C, E, J, K and L.
Reason for in-correct options:
The remaining compounds do not contain delocalized electrons since the conjugated system is absent.
Hence, the in-correct options are D, F, G, H, I, J, and K.
The compounds containing delocalized electrons were predicted.
Want to see more full solutions like this?
Chapter 7 Solutions
Essential Organic Chemistry, Global Edition
- Hi, I need help writing these equations. A little confused. I think a is CH3CHCH2CH3+H20. b.is CH3C(CH3)2CH2CH2CH2CH3+Br is that correct? thanksarrow_forward1. Which of the followings will be deflected to the LEAST extent? a. +CH2CH3 b. +CH3 c. +CN d. +OHarrow_forwardWhich term best describes the following rxn? A. regioselective B. stereoselective C. both D. neitherarrow_forward
- Plane-polarized light is transmitted through a chamber that contains a single enantiomer and rotates to the right. Plane-polarized light passed through a chamber containing the other enantiomer would rotate ________ A. to the right B. not at all C. in a way that cannot be predicted D. to the left A 1:1 mixture of the enantiomers would rotate ______ A. to the right B. not at all C. in a way that cannot be predicted D. to the leftarrow_forwardDoes 2,3-dichlorobutane have optical activity?(1). No.(2). Only in solution.(3). Yes, in any state (solid, liquid and gas).(4). It depends on the stereoisomer we are referring to.arrow_forwardwhy does n = t1/t2?arrow_forward
- Based on the image. What is 1. The anti-conformer of hexane in Newman projection. 2. The equivalent structure of LAM in sawhorse representation. 3.arrow_forwardWhich group in the following pair is assigned the higher priority in R,Snomenclature −CD3, −CH3arrow_forwardA) Draw the structure of any aldohexose in the pyranose ring form. B) Draw the structure of the anomer of the aldohexose you drew above. C) How many asymmetric carbons (chiral centers) does each of these structures have? D) How many stereoisomers of the aldohexoses you drew are theoretically possible?arrow_forward
- 16) how many chiral carbons are in this moleculearrow_forwardHow many aldooctoses are possible? How many d-aldooctoses are possible?arrow_forwardWhich of the do/does not rotate the plane of polarized light? a. 3-methyloctane b. (-)-lactic acid c. None of the choices d. (+)-glucose e. (±)-sucrosearrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
