
(a)
Interpretation:
The major product of the reaction has to be identified.
Concept introduction:
Carbocation: Carbocation is a positive charged species and vital intermediate in
Hydrobromination:
A hydrobromination reaction is one of the electrophilic additions to
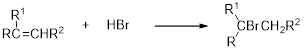
(b)
Interpretation:
The major product of the reaction has to be identified.
Concept introduction:
Carbocation: Carbocation is a positive charged species and vital intermediate in organic synthesis and its movement depends on the stability of the intermediate and the product formation.
Hydrobromination:
A hydrobromination reaction is one of the electrophilic additions to alkenes to yield the corresponding bromo alkanes. In this reaction the bromine atom adds to the double bond carbon which is having lesser number of hydrogen or more substituted carbon (Markovnikov's rule).
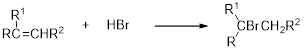
Want to see the full answer?
Check out a sample textbook solution
Chapter 7 Solutions
Essential Organic Chemistry, Global Edition
- Please provide the steps that each carbonation will undergo to form a carbonation that is more stable. Use arrows to explain the steps and provide the product for each.arrow_forwardWrite the structure of the principal organic product of each reactionarrow_forwardWhat is the major product formed from this reactionarrow_forward
- By taking into account electronegativity differences, draw the products formed by heterolysis of the carbon–heteroatom bond in each molecule. Classify the organic reactive intermediate as a carbocation or a carbanion.arrow_forwardSynthesize the product shown from the starting materials, including intermediates formed at each steparrow_forwardParts A and B are the images, and Part C has the same reactant but H2O and H2SO4 as reagents. Draw the product for each reaction.arrow_forward
- In the following series of reactions, write the product (s) or the reagents needed to obtain the indicated product.arrow_forwardDraw the two major products obtained in the reaction shown. CH2Cl2 Br2 (one equivalent)arrow_forwardIdentify the best reagents to complete the following reaction. HO, CIarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





