
Study Guide for Campbell Biology
11th Edition
ISBN: 9780134443775
Author: Lisa A. Urry, Michael L. Cain, Steven A. Wasserman, Peter V. Minorsky, Jane B. Reece, Martha R. Taylor, Michael A. Pollock
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 7, Problem 8TYK
Use the following U-tube setup to answer questions 6 through 8.
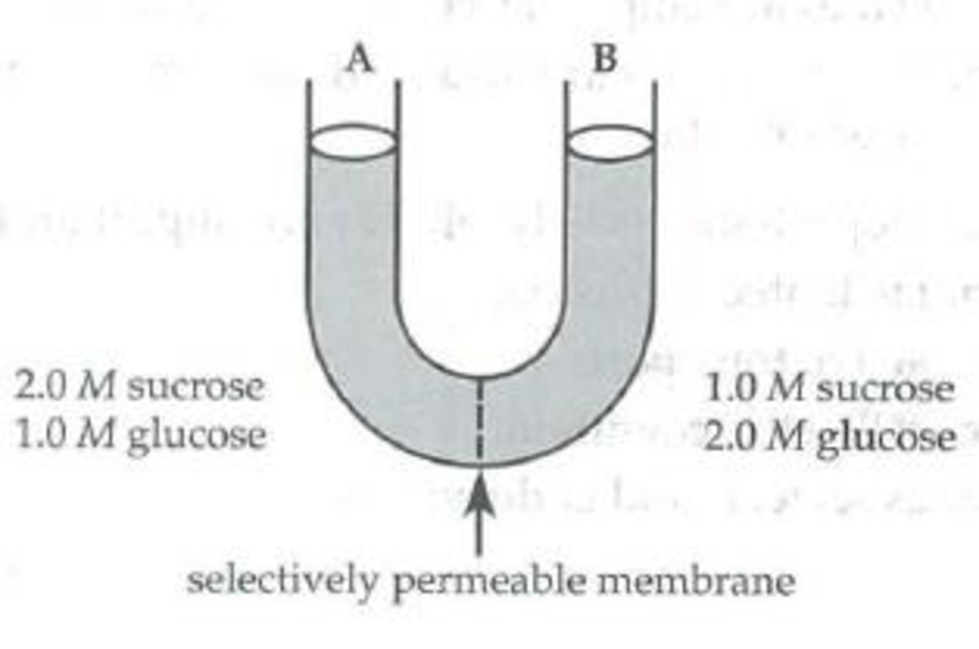
The solutions in the two arms of this U-tube are separated by a membrane that is permeable to water and glucose but not to sucrose. Side A is filled with a solution of 2.0 M sucrose and 1.0 M glucose. Side B is filled with 1.0 M sucrose and 2.0 M glucose.
After the system reaches equilibrium, what changes can be observed?
- a. The water level is higher in side A than in side B.
- b. The water level is higher in side B than in side A.
- c. The molarity of glucose is higher in side A than in side B.
- d. Both a and c have occurred.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
The beaker is divided into two compartments (C and D) that has equal volumes of solution separated by an artificial membrane that has a pore size of 23 Å (Angstrom). Explain the movement of solute after eight hours if comparment C has 17 % sucrose while the compartment D has 2 % sucrose (diameter of sucrose molecule = 9 Å (Angstrom))
The left beaker contains 9mM NaCl and 9mM glucose solution.The right beaker contains distilled water.The molecular weight of NaCl is 58.44 g/mol. The molecular weight of glucose is 180.16 g/mol. The membrane located between the two beakers is impermeable to NaCl and glucose. The membrane is permeable to H2O. What will occur across the membrane?
An artificial cell consisting of an aqueous solution enclosed in a selectively permeable membrane (but with no cell wall) is immersed in a beaker containing an aqueous solution. The outside environment concentration consists of 0.01 M glucose and the inside of the cell has a concentration of 5.0 M glucose. The plasma membrane is permeable to water and monosaccharides, but impermeable to the disaccharides. Complete the following for the image below:
Will the artificial cell become lyse, shrivel, or stay the same?
lyse
shrivel
stay the same
Chapter 7 Solutions
Study Guide for Campbell Biology
Ch. 7 - Label the components in the following diagram of a...Ch. 7 - a. Cite some experimental evidence that indicates...Ch. 7 - List the six major functions that membrane...Ch. 7 - What types of molecules have difficulty crossing...Ch. 7 - A solution of 1 M glucose is separated by a...Ch. 7 - a. What osmotic problems does the freshwater...Ch. 7 - Prob. 7IQCh. 7 - Prob. 8IQCh. 7 - a. How is cholesterol transported into human...Ch. 7 - Create a concept map to illustrate your...
Ch. 7 - The following diagram illustrates passive and...Ch. 7 - If a single layer of phospholipids coats the water...Ch. 7 - Glycoproteins and glycolipids are important for a....Ch. 7 - Prob. 3TYKCh. 7 - Prob. 4TYKCh. 7 - Prob. 5TYKCh. 7 - Use the following U-tube setup to answer questions...Ch. 7 - Use the following U-tube setup to answer questions...Ch. 7 - Use the following U-tube setup to answer questions...Ch. 7 - How much work is expended in diffusion? a. an...Ch. 7 - Prob. 10TYKCh. 7 - Prob. 11TYKCh. 7 - Water passes quickly through cell membranes...Ch. 7 - Facilitated diffusion of ions across a cellular...Ch. 7 - Prob. 14TYKCh. 7 - Which of the following describes cotransport? a....Ch. 7 - Prob. 16TYKCh. 7 - An animal cell moves potassium against its...Ch. 7 - Prob. 18TYKCh. 7 - Prob. 19TYKCh. 7 - In response to low blood pressure, the adrenal...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biology and related others by exploring similar questions and additional content below.Similar questions
- An artificial cell consisting of an aqueous solution enclosed in a selectively permeable membrane (but with no cell wall) is immersed in a beaker containing an aqueous solution. The outside environment concentration consists of 0.01 M glucose and the inside of the cell has a concentration of 5.0 M glucose. The plasma membrane is permeable to water and monosaccharides, but impermeable to the disaccharides. Complete the following for the image below: Is the glucose going down or against its concentration gradient? Is the movement of the solute in the cell going out of the cell? Down...Yes Down...No Against...Yes Against...Noarrow_forwardfor the following scenario, DRAW the situation depicted, INDICATE which solution is hypertonic, hypotonic or isotonic (if applicable), and then show with ARROWS which way water will move. then write your response in complete sentences, thoroughly explaining and justifying your arrows. a. ouch! sore throat! it is so swollen that it is sore and it is difficult to swallow. and you have an oral presentation today. better gargle with salt water to reduce the swelling! explain why this age old technique really works, by using the concept of osmosisarrow_forwardShown below are cells (colored) that were recently placed into a beaker containing a clear solution For each scenario, indicate whether movement of the molecule into the cell will occur using facilitated diffusion or active transport. А. B. 125mM 20mM fructose glucose 85MM 35mM fructose glucose OA= facilitated diffusion; B= facilitated diffusion OA= active transport; B= active transport O A= active transport; B= facilitated diffusion A= facilitated diffusion; B= active transportarrow_forward
- An artificial cell consisting of an aqueous solution enclosed in a selectively permeable membrane (but with no cell wall) is immersed in a beaker containing an aqueous solution. The outside environment concentration consists of 0.01 M glucose and the inside of the cell has a concentration of 5.0 M glucose. The plasma membrane is permeable to water and monosaccharides, but impermeable to the disaccharides. Complete the following for the image below: Is the solution inside the cell isotonic, hypotonic, or hypertonic? Is the glucose going down or against its concentration gradient? Draw solid arrow to indicate the movement of the solute into or out of the cell. Draw a dashed arrow to show the movement of water. Will the artificial cell become lyse, shrivel, or stay the same?arrow_forwardA student designed an experiment to test whether different concentration gradients affect the rate of diffusion through dialysis tubing. Four different solutions (0% NaCl, 1% NaCl, 5% NaCl, and 10% NaCl) were tested under identical conditions. Twenty mL of each solution was placed into separate dialysis tubing that is permeable to Na+, Cl-, and water. Each bag was placed in a separate beaker and covered with distilled water. The concentration of NaCl in the water outside of each bag was measured at 40-second intervals. The graph below shows the results from the 5% bag: Concentration of NaCl Outside of Dialysis Bags Concentration of NaCl (mg/L) 400 320 240 160 80 0 40 80 Time(seconds) 120 5% Solution 160 a. Describe the process of diffusion through the dialysis tubing. b. If you were to design a different experiment testing rates of diffusion, identify experimental procedures. c. Analyze that data in the graph and describe the results. Provide reasoning to justify your answer. d.…arrow_forwardA red blood cell with an intracellular fluid (ICF) concentration of 280 mmol/L, is placed into an environment were the concentration of the extracellular fluid (ECF) is 305 mmol/L. As a result, water will move across the cell/plasma membrane. a) Name the process by which water moves b) Describe the overall direction of movement of the water. Answer choices: (i) From outside the cell to inside the cell (ii) From inside the cell to outside the cell, or (iii) Equally, into and out of the cell iii Explain your reasoning for your answer to part (c). 1In your answer you must describe the environment on both sides of the membrane, and use 3 of the following 4 terms correctly; hypertonic, hypotonic, lower solute concentration, higher solute concentrationn (2pts -Correct use of biological terminology, and correct spelling is essential. Edit View Insert Format Tools Table 12pt v Paragraph BIUA ...arrow_forward
- In this part of the assignment, assume you have carried out the microscopic examination of the effects of osmosis in plant cells experiment The image in Figure 4 is of Elodea canadensis leaf cells in isotonic solution (normal saline). The image in Figure 5 is of Elodea canadensis leaf cells in 10% sucrose solution. The image in Figure 6 is of Elodea canadensis leaf cells in distilled water. a. Determine cell size and vacuole size. b. Calculate the percentage change in vacuole size for each treatment compared to the control. Use the formula: Experimental Size - Control Size/Control Size c. Based on these results, explain the tonicity of each treatment using experimental results to support your answer as would appear in the discussion section of a formal report (maximum four (4) lines). d. Was the percentage change in vacuole size for the experimental conditions relative to the control of similar magnitude? If the change was not of the same magnitude, explain the difference in effect…arrow_forwardIn the experiment showing osmosis through a semipermeable membrane, 3 corn syrup mixtures are used. If equal volumes of two solutions (10% corn syrup and 30% corn syrup) were placed in two beakers that were separated by a semipermeable membrane, permeable only to water, and let sit for 1 hour, what would be the expected concentration/s of the two solutions at the end of the time period? Explain using solute concentration, water concentration etc.arrow_forwardIn an investigation of osmosis, apple cores were submerged in different molarity solutions to determine the unknown osmolarity of the apple. To determine the molarity five uniform apple cores were massed before submerging them in different molarity solutions. After 24 hours the apple cores were massed again. Then the percent change in mass was determined for the five uniform apple cores in each molarity solution. c) Describe the environmental conditions taking place in each of the six solutions with the applesarrow_forward
- a. Define diffusion. Is diffusion a passive or an active process? Explain how solute moves in this way. b. Use your knowledge of diffusion to explain what happened over time when you observed a crystal of methylene blue dropped into a beaker of water. Be sure to use equilibrium in your explanation. c. Explain your observations over time after a drop of methylene blue and a drop of potassium permanganate were placed in the agar. What factors affect the rate of diffusion? d. What are tissues? Name the four major types of tissues in the human body. e. Name the three primary germ layers. Name the primary germ layers from which epithelial tissues and connective tissues develop. f. List general characteristics that all epithelial tissues have in common. g. Name the functions of epithelial tissue. Which type of epithelial tissue is found in areas that need protection? h. Know how to identify simple squamous epithelium, simple cuboidal epithelium, simple columnar…arrow_forwardAnswer the following questions based on the graph: a) Which of the two molecules that are the same size will passively diffuse across a cell membrane quicker? Why? b) Which type of transport method would glucose use? Why? Figure 1: Relative Sizes of Molecules Diffusing Across a Cell Membrane Substance Tested water oxygen glycerol glucose alcohol carbon dioxide Graph 0 50 = 150 100 Relative Size of Molecules 200arrow_forwardFor the following scenario you must draw the situation depicted, indicate which solution is hypertonic, hypotonic or isotonic (as applicable), and then show with arrows which way water will move. Then you must write your response in complete sentences, thoroughly explaining and justifying your arrows. a. Ouch! Sore throat! It is so swollen that it is sore and it is difficult to swallow. And you have an oral presentation today. Better gargle with salt water to reduce the swelling! Explain why this age old technique really works, by using the concept of osmosis.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Human Physiology: From Cells to Systems (MindTap ...BiologyISBN:9781285866932Author:Lauralee SherwoodPublisher:Cengage Learning
Human Physiology: From Cells to Systems (MindTap ...BiologyISBN:9781285866932Author:Lauralee SherwoodPublisher:Cengage Learning

Human Physiology: From Cells to Systems (MindTap ...
Biology
ISBN:9781285866932
Author:Lauralee Sherwood
Publisher:Cengage Learning
The Cell Membrane; Author: The Organic Chemistry Tutor;https://www.youtube.com/watch?v=AsffT7XIXbA;License: Standard youtube license