
Macroscale and Microscale Organic Experiments
7th Edition
ISBN: 9781305577190
Author: Kenneth L. Williamson, Katherine M. Masters
Publisher: Brooks Cole
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 7, Problem 8Q
Interpretation Introduction
Interpretation:
A flow chart that depicts the separation procedure used for separation of mixture of toluic acid,
Concept introduction:
Separation of a mixture that contains an acidic, basic and neutral substance involves acid-base extraction technique.
The structure of toluic acid is as follows:
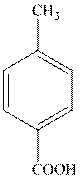
The structure of
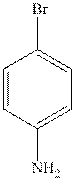
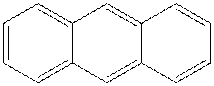
The structure of anthracene is as follows:
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
Draw a flow sheet to show how you would separate the components of a mixture containing an acid substance, toluic acid, a basic substance, p-bromoaniline, and anthracene, a neutral substance.
Draw a scheme to show how one would separate a mixture of naphthalene and aniline (Hint: aniline is an organic base that reacts with HCl(aq) to form a salt).
Write the chemical equation between acetic acid and sodium hydroxide.
Chapter 7 Solutions
Macroscale and Microscale Organic Experiments
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Write the chemical equation for a pentanoate ion acting as base when it reacts with hydrochloric acid (HCI). Which is formula of this rule of reaction?arrow_forwardIdentify some of the detergents used in shampoos and dishwashing liquids. Are they primarily anionic, neutral, or cationic detergents?arrow_forwardWrite the molecular equation for the reaction of CH3COOH and NaOH. Write the net ionic equation for the reaction of CH3COOH and NaOH.arrow_forward
- Salicylic acid is added into a test tube, followed by methanol and sulfuric acid as the catalyst. It is heated for 10-15 minutes and poured into a beaker with crushed ice. This is the esterification of Oil of wintergreen. Write the complete reaction equation and describe the odor of the reactants (salicylic acid and methanol) and the products (oil of wintergreen).arrow_forwardWrite the balanced chemical equation for the reaction of oxalic acid dihydrate with sodium hydroxide.arrow_forwardAlkaloids are basic nitrogen-containing compounds of plant origin, many of which are physiologically active when administered to humans. Ingestion of coniine, isolated from water hemlock, can cause weakness, labored respiration, paralysis, and eventually death. Coniine is the toxic substance in the “poison hemlock” used in the death of Socrates. In small doses, nicotine is an addictive stimulant. In larger doses, it causes depression, nausea, and vomiting. In still larger doses, it is a deadly poison. Solutions of nicotine in water are used as insecticides. Cocaine is a central nervous system stimulant obtained from the leaves of the coca plant.Classify each amino group in these alkaloids according to type (primary, secondary, tertiary, aliphatic, aromatic, heterocyclic).arrow_forward
- Write the chemical equation for the acid dissociation of acetaminophen, C8H9O2N. Write the Ka expression for the acid dissociation of acetaminophen.arrow_forwardwhat would the chemical reaction be for the process of aspirin when combining salicylic acid, phosphoric acid, and acetic anhydridearrow_forward18-23 Characterize the structural features necessary to make a good synthetic detergent.arrow_forward
- 18-17 Hexanoic (caproic) acid has a solubility in water of about 1 g/100 mL water. Which part of the molecule contributes to water solubility, and which part prevents solubility?arrow_forwardWhat are some medical applications of tannic acid?arrow_forwardA) Write an equation for the synthesis of aspirin and give the names of the reactants and products. B) How many aspirin tablets can I prepare from 261 grams of acetylsalicylic acid (ASA)? Remember that the commercial aspirin tablet contains 325 mg ASA. C) What functional group of aspirin causes irritation to the lining of the stomach?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning

Macroscale and Microscale Organic Experiments
Chemistry
ISBN:9781305577190
Author:Kenneth L. Williamson, Katherine M. Masters
Publisher:Brooks Cole

Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:9780078746376
Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:Glencoe/McGraw-Hill School Pub Co

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning