
Concept explainers
Interpretation:
Equations that describe the extraction procedure of acetaminophen from an organic solvent such as ether should be written.
Concept introduction:
Acetaminophen is commonly known as paracetamol.It is used as an analgesic and antipyretic.IUPAC name of paracetamol is
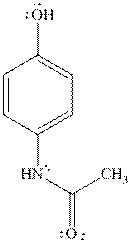
Extraction of drug like acetaminophen can be done withan appropriate choice of solvents. There are several techniques for the extraction ofthe drug. The commonly used methods include column chromatography and liquid-liquid extraction that is carried out in the presence of water and ether as the solvents. The difference in densities of water and ether allow them to form separate layers in a separatory funnel and thus allows the effective separation and isolation of caffeine.Experimental results reveal that paracetamol is soluble in acetone, ethanol.
Trending nowThis is a popular solution!

Chapter 7 Solutions
Macroscale and Microscale Organic Experiments
- Is Tripamide soluable in waterarrow_forwardWhen comparing the structures of aspirin and acetaminophen, why can acetaminophen exist in a liquid state (i.e. childrens drinkable tylenol) and aspirin cannot? Does it have to do with the carboxylic acid? The question says that liquid aspirin cannot be made. Why would that be?arrow_forwardWrite the products of the reaction of diphenhydramine (a base) with the acid HCl shown below. Are the reactants or products more soluble in water? Briefly explain.arrow_forward
- why Acetanilide after cooling back at room temperature forms a crystal?arrow_forwardExplain the solubility behavior of aniline, diethylamine and N,N-diethylaniline in water.arrow_forwardWrite the structure of benzylamine hydrochloride in two different ways, and name the hydrochloride as an ammonium salt.arrow_forward
- Discuss the differences among the following compounds by drawing their structures: Acetylsalicylic acid, Acetaminophen, Ibuprofen, Paracetamol and Phenacetin.arrow_forwardWhat is the white solid that forms when α-phenylethylamine comes in contact with carbon dioxide? Write an equation for its formulation.arrow_forwardDraw several molecules of methanamide and show the intermolecular hydrogen bonding that account for the amide’s very high boiling point.arrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning


