
Concept explainers
Piperine, the active ingredient in black pepper, has this Lewis structure.
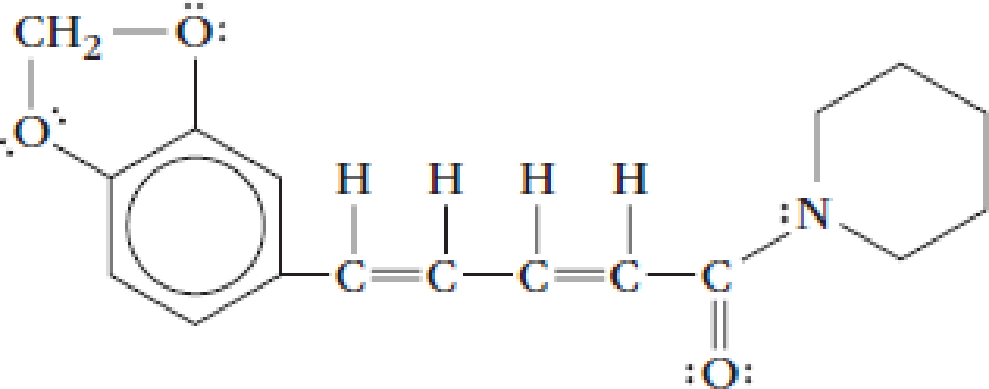
- (a) Give the values for the indicated bond angles.
- (b) What is the hybridization of the nitrogen?
- (c) What is the hybridization of the oxygens?
(a)
Interpretation:
The indicated bond angles has to be given.
Concept Introduction:
Bond angle is the angle between two bonds of a molecule and it is determined based on the electron-domain geometry.
Explanation of Solution
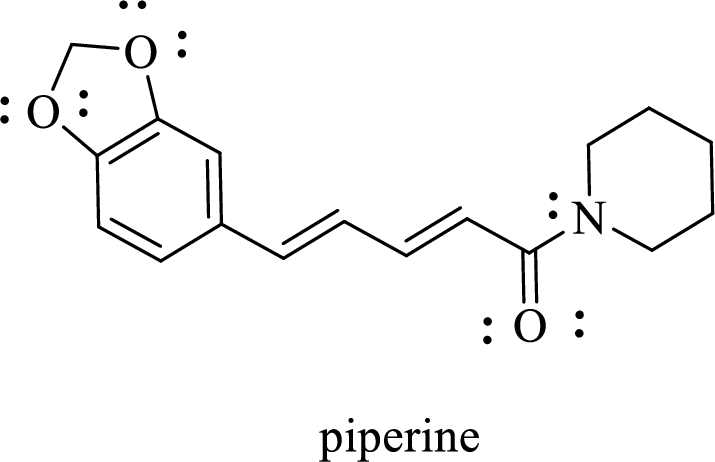
The given structure of Piperine is given below:
 .
.
The bond angles to be determined are given below:
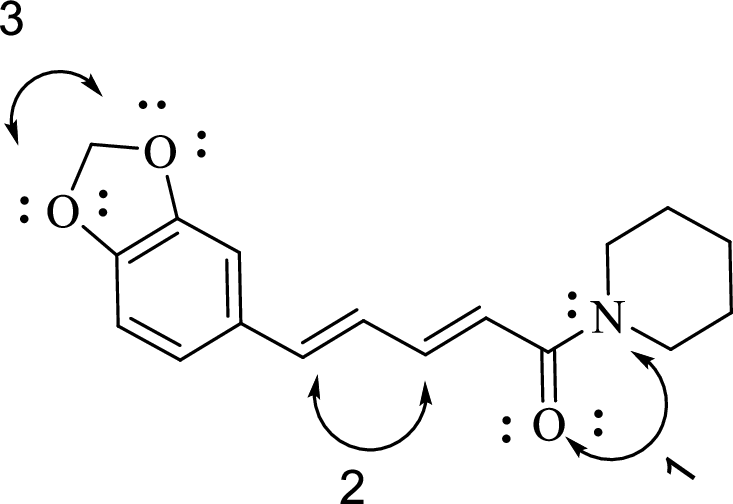 .
.
Consider the
Consider the
Consider the
(b)
Interpretation:
The hybridization for nitrogen atom in the molecule has to be designated.
Concept Introduction:
Hybridization is the mixing of valence atomic orbitals to get equivalent hybridized orbitals that having similar characteristics and energy.
Geometry of different types of molecule with respect to the hybridizations are mentioned below,
Explanation of Solution
The given structure of Piperine is shown below:
 .
.
Consider the nitrogen atom. There will be four electron regions around nitrogen atom. It is a type of
Consider the right carbon. There will be three electron regions in the molecule and hence the electron-region geometry will be triangular planar. For a molecule having tetrahedral geometry, the hybridization will be
(c)
Interpretation:
The hybridization for oxygen atoms in the molecule has to be designated.
Concept Introduction:
Refer to (b).
Explanation of Solution
The given structure of Piperine is shown below:
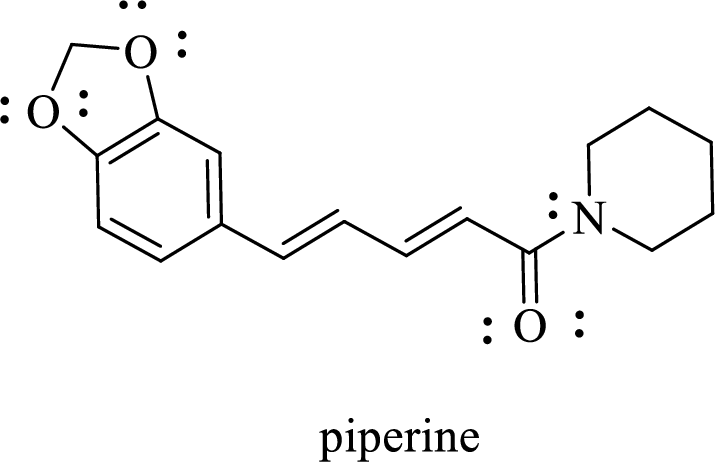 .
.
Consider the nitrogen atom. There will be four electron regions around nitrogen atom. It is a type of
Consider the oxygen atoms with single bonds. There will be four electron regions around both oxygen atoms. They are a type of
Consider the double bonded oxygen atom. There will be three electron regions around oxygen atom. It is a type of
Want to see more full solutions like this?
Chapter 7 Solutions
Chemistry: The Molecular Science
- Draw the Lewis structure of azide (N₃⁻) and then determine the hybridization of the central atom.arrow_forwardConsider the molecules H3O+, HCHO, IF7Draw the Lewis structures, hybridizations of central atom, molecular geometry and degree of angle is expected.arrow_forwardThe existence of compounds of the noble gases was once a great surprise and stimulated a great deal of theoretical work. Sketch the molecular orbital energy level diagram for XeF and deduce its ground-state electron configurations. Is XeF likely to have a shorter or longer bond than XeF+?arrow_forward
- 4.What is the most important information needed in order to predict the hybridization of orbitals on a central atom?arrow_forwardWhat is the molecular structure of C2H5OH, specifiy the shape and hybridization about each "central" atom? Please include the drawing of the molecular structure.arrow_forwardAspirin, or acetylsalicylic acid, has the formula C9H8O4 and the skeleton structure (a) Complete the Lewis structure and give the number of bonds and bonds in aspirin. (b) What is the hybridization about the CO2H carbon atom (colored blue)? (c) What is the hybridization about the carbon atom in the benzene-like ring that is bonded to an oxygen atom (colored red)? Also, what is the hybridization of the oxygen atom bonded to this carbon atom?arrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning

