
(a)
Interpretation: The electron sublevel starts to fill after the completion of 5s sublevel should be determined.
Concept Introduction:
The rules for the allowed quantum numbers combinations are as follows:
- All the three quantum numbers ( n, l and m ) describes the orbital of an atom and they are integers.
- The principal quantum number, n value cannot be zero. Thus, the values allowed for the principal quantum number are 1, 2, 3, 4, and so on.
- The value of angular quantum number, l can be between 0 to n-1. Thus, if value of n is equal to 3 the value of l can be 0, 1 or 2.
- The value of magnetic quantum number, m can be between − l to +l . Thus, if value of l is equal to 2, m can be wither -2, -1, 0, +1, or +2
For same number of principal quantum number, an orbital form a shell. The first character denotes the shell and the second identifies the sub-shell.
Here, for s orbital value of
The relative energy of orbitals is represented as follows:
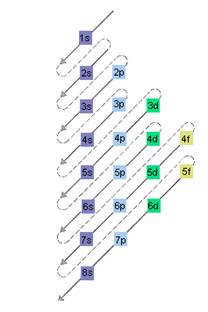
(b)
Interpretation: The electron sublevel starts to fill after the completion of 4d sublevel should be determined.
Concept Introduction:
The rules for the allowed quantum numbers combinations are as follows:
- All the three quantum numbers ( n, l and m ) describes the orbital of an atom and they are integers.
- The principal quantum number, n value cannot be zero. Thus, the values allowed for the principal quantum number are 1, 2, 3, 4, and so on.
- The value of angular quantum number, l can be between 0 to n-1. Thus, if value of n is equal to 3 the value of l can be 0, 1 or 2.
- The value of magnetic quantum number, m can be between − l to +l . Thus, if value of l is equal to 2, m can be wither -2, -1, 0, +1, or +2
For same number of principal quantum number, an orbital form a shell. The first character denotes the shell and the second identifies the sub-shell.
Here, for s orbital value of
The relative energy of orbitals is represented as follows:
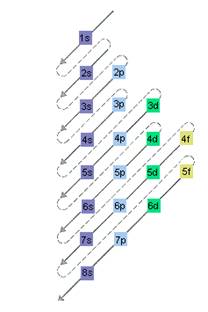
(c)
Interpretation: The electron sublevel starts to fill after the completion of 4f sublevel should be determined.
Concept Introduction:
The rules for the allowed quantum numbers combinations are as follows:
- All the three quantum numbers ( n, l and m ) describes the orbital of an atom and they are integers.
- The principal quantum number, n value cannot be zero. Thus, the values allowed for the principal quantum number are 1, 2, 3, 4, and so on.
- The value of angular quantum number, l can be between 0 to n-1. Thus, if value of n is equal to 3 the value of l can be 0, 1 or 2.
- The value of magnetic quantum number, m can be between − l to +l . Thus, if value of l is equal to 2, m can be wither -2, -1, 0, +1, or +2
For same number of principal quantum number, an orbital form a shell. The first character denotes the shell and the second identifies the sub-shell.
Here, for s orbital value of
The relative energy of orbitals is represented as follows:
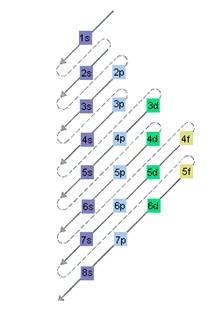
(d)
Interpretation: The electron sublevel starts to fill after the completion of 5p sublevel should be determined.
Concept Introduction:
The rules for the allowed quantum numbers combinations are as follows:
- All the three quantum numbers ( n, l and m ) describes the orbital of an atom and they are integers.
- The principal quantum number, n value cannot be zero. Thus, the values allowed for the principal quantum number are 1, 2, 3, 4, and so on.
- The value of angular quantum number, l can be between 0 to n-1. Thus, if value of n is equal to 3 the value of l can be 0, 1 or 2.
- The value of magnetic quantum number, m can be between − l to +l . Thus, if value of l is equal to 2, m can be wither -2, -1, 0, +1, or +2
lFor same number of principal quantum number, an orbital form a shell. The first character denotes the shell and the second identifies the sub-shell.
Here, for s orbital value of
The relative energy of orbitals is represented as follows:
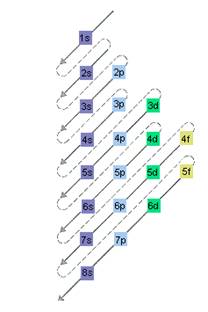
Want to see the full answer?
Check out a sample textbook solution
Chapter 5 Solutions
Basic Chemistry
- none of the above; there is no such particle QUESTION 5 Orbital shells of an atom: O contain electrons and are found in the nucleus O contain protons and are found in the nucleus O contain electrons and surround the nucleus O contain protons and surround the nucleus QUESTION 6 Suppose an oxygen atom has 8 protons and 9 neutrons. How many electrons does it have? 9. 17 Click Save and Submit to save and submit. Click Save AL Answers to save all answers. archarrow_forward4.52 Identify the elements that have the following electron arrangements: Energy Level 4 a. 2 b. 8 6. C. 4 d. 8. 1. e. 8. 5.arrow_forward1. By using the noble gas notation, write down the ground state electron configuration of the following elements. (5)1.1 Carbon1.2 Phosphorus 1.3 Scandium 1.4 Tungsten 1.5 Antimonyarrow_forward
- The effective nuclear charge of a nucleus is determined from a shielding factor. The shielding factor depends on the number of shielding electrons in the atoms' electronic structure. How many "shielding electrons" are present in the ground state of the element In (No. 49) Number of "shielding electrons": Answerarrow_forward6 of 11 I Review Constants Periodic Table Part C out ars. ying lue Consider the following equivalent expressions: 3 13 3 and 12 a 8 What are the values of a and b? Enter the value of a followed by the value of b, separated by a comma. - get the ons) ple, Submit Previous Answers Request Answer ") is X Incorrect; Try Again; 4 attempts remaining Part D Complete previous part(s)arrow_forward4.74 Which statements completed with a-d will be true and which will be false? An atom of Pb compared to an atom of C has a larger (greater) b. ionization energy d. metallic character a. number of neutrons c. atomic sizearrow_forward
- 12:13 Question 8 of 17 Submit Rank the following atoms in order of decreasing size (i.e., largest to smallest): Rb, F, Mg, B, N. A) Rb > Mg >B >N > F B) N > F>B > Mg > Rb C) Rb > Mg > F >N> B D) Mg > Rb >B >N > F E) Rb > Mg >N > B > F Tap here or pull up for additional resourcesarrow_forward1.1 What is the difference between the core electrons and valence electrons ? Why do we emphasize the valence electrons in an atom when discussing atomic properties? 1.2 Ranking the following in order of increasing atomic radius O, S and F? 1.3 Explain why sizes atoms change when proceeding across a period of the periodic table.arrow_forwardComplete the followingequations: 32p with an atomic number of 15 splits into _______ and an electron and varrow_forward
 Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning

