
Consider the wall section shown in Fig. -4a, construction 1, and estimate the temperature of the inside surface of the concrete block at the furring. The outdoor temperature is 1 F (—17 C) and the inside temperature is 72 F (22 C) with a relative humidity of 45 percent. Would you recommend a vapor retardant? If so, where would you place it? Explain.
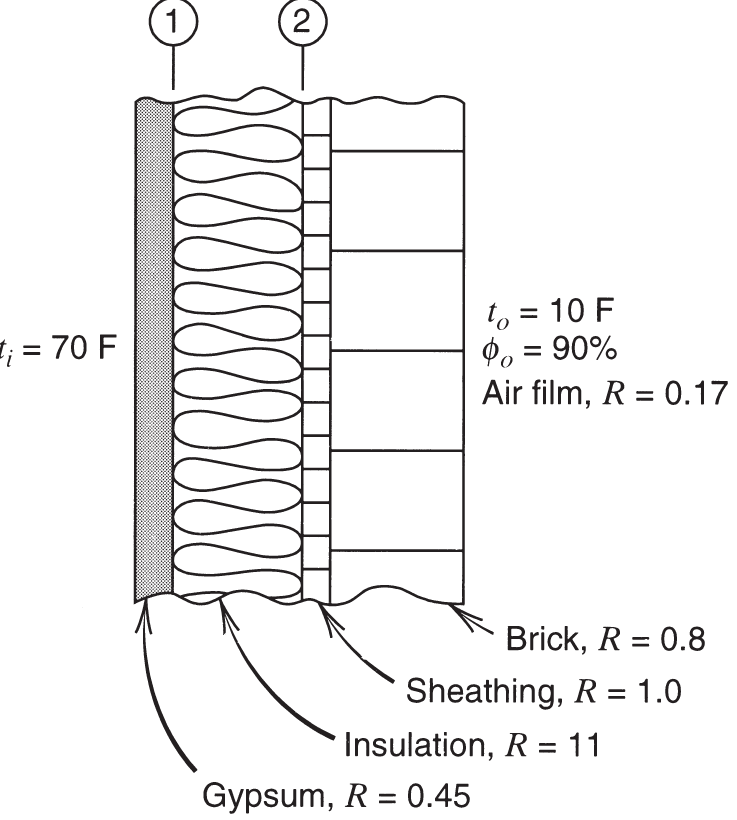
Figure 5-10 Wall section for Problem 5-36.
Want to see the full answer?
Check out a sample textbook solution
Chapter 5 Solutions
Heating Ventilating and Air Conditioning: Analysis and Design
Additional Engineering Textbook Solutions
Vector Mechanics for Engineers: Statics, 11th Edition
Introduction To Finite Element Analysis And Design
Mechanics of Materials (10th Edition)
Shigley's Mechanical Engineering Design (McGraw-Hill Series in Mechanical Engineering)
Statics and Mechanics of Materials (5th Edition)
DeGarmo's Materials and Processes in Manufacturing
- The Occupational Safety and Health Administration has set a Permissible Exposure Limit of 1000 parts per million (ppm) of acetone. You are an industrial hygienist doing personal sampling on a worker who is supervising a production process that uses acetone. The upcoming exposures can be measured: 700 ppm for three hours and 1300 ppm for two hours. 3 hours at 900 ppm What is the TWA as calculated? What advice would you provide plant management regarding acetone exposure and PEL adherence?arrow_forwardPROBLEM: IIC - 12 BOOK: ENGINEERING THERMOFLUIDS, M. MASSOUDarrow_forwardWhich statements about case hardening are correct? The process makes it possible to harden local areas of the component. The process promotes a fine-grain structure due to relatively low heating temperatures. Case hardening is particularly useful for austenitic steels Case hardening is well suited for small workpieces. During case hardening, the workpiece is specifically enriched with carbon and nitrogen. After in-diffusion, the workpiece is tempered with the carburized gas medium:arrow_forward
- The Occupational Safety and Health Administration has established a Permissible Exposure Limit of 200 parts per million (ppm) for toluene. As an industrial hygienist, you are doing personal sampling on a worker who is in charge of a production process that uses toluene. The upcoming exposures can be measured: During one hour, 500 ppm 100 ppm for four hours and 225 ppm for three What is the TWA as calculated? What advice would you provide plant management regarding toluene exposure and PEL adherence?arrow_forwardMetal spheres, 10 mm in diameter, are to be annealed by heating them to 827°C then allowing them to cool slowly in air at 27°C to the point where they are in thermal equilibrium with the air. 2000 balls are annealed in one hour’s time. What is the total rate of their heat transfer if they are made of steel, lead, or copper? a.Sketch the problem. b.Draw lines identifying the control volume, or control mass. c.Identify the states with numbers, letters, or descriptions such as “in” and “out”. d.Write down the knowns and unknowns. e.Identify what is being asked for. f.State all assumptions.arrow_forwardhnen't For 1 mole of a gas, the van der Waals equation is where R is the gas constant (0.0821 L atm K mol ') and 7 is the Kelvin temperature 1The constants a and h are constants particular to a given gas and correct for the attractive forces between gas molecules, and for the volume occupied by the gas molecules, respectively. For methane (CH), the constants are a 2.253 L'atm and b4.278 x 10 L. Using the rearranged form of the van der Waals equation RT V -b v? calculate the pressure of 1 mole of methane as a function of container volume 0°C (273 K) at suitable volumes from 22.4 L to 0.05 L. Use one of the at custom functions described in this chapter to calculate the first and second derivatives of the P-V relationship. Compare with the exact expressions dP RT 2a dV (v - b)?v3 d? P 2RT 6a dv? (V -b)arrow_forward
- Refer to Table 3.2 in the lecture and take the data for air, water (steam), and four other gases listed in the table. Plot the data in a graph with the thermal conductivity on the vertical axis and the temperature range from 200 K to 600 K on the horizontal axis. Use colors or different dashed lines to make the graph presentable and understandable. The graph should look like the graph in Figure 3.1 in the lecture. Provide a short explanation of the graph in three to five sentences.arrow_forwardVolumetric expansion coefficients of simple materials are often well cataloged. However, the thermal expansion coefficient B of a human body is less well known. This could affect the human body's specific gravity and, therefore, measurements of its body-fat ratio. Suppose that a human body of weight wo on dry land is placed on a scale while completely immersed in formaldehyde of temperature T1- Once the temperature increases by AT, the scale reading drops by Aw. Derive an expression for ß in terms of AT, wo, and Aw by assuming that the ratio of the density of formaldehyde ef and the initial density of the body Po is R = Pf/pb. Assume also that pf does not change when heated. B = %3D If the body weighs 237.2 lb on dry land and his weight reading lowers by 0.237 lb when the formaldehyde is heated from 63.40 °F to 82.80 °F, calculate the coefficient of volume expansion of the body.* Assume R = 0.820. 1/°C B =arrow_forwardjust i need solution for task 2 Task1 Using the following gases: hydrogen, carbon monoxide, carbon dioxide, and oxygen determine the specific heats at constant volume and constant pressure maintained at different temperatures :20 ºC, 25 ºC, 30 ºC, and 40 ºC. What is the effect of temperature on specific heat for different gases. You can present your data in a table or graph since you have different gases and temperatures. Calculate the gas constant for different gases maintained at different temperatures and compare your results to the published data. A fixed mass of an ideal gas is heated from 50 to 80 C at a constant pressure of (a) 1 atm and (b) 3 atm. For which case do you think the energy required will be greater? Why? A fixed mass of an ideal gas is heated from 50 to 80 C (a) at constant volume and (b) at constant pressure. For which case do you think the energy required will be greater? Why? Task 2 A frictionless piston fitted inside a cylinder containing gas at a…arrow_forward
- PROBLEM: IIC-21 BOOK: ENGINEERING THERMOFLUIDS, M. MASSOUDarrow_forwardProblem 7 Can of Gas A can with volume V has N molecules of nitrogen contained inside. A plug at the top has mass m and cross-sectional area A. The plug can withstand a force of 2mg before it would pop loose. The pressure of the outside air is Po. (a) Draw a free-body diagram of the plug. (b) Assuming the outside air and gas inside the container have the same temperature, at what outdoor temperature would the plug pop loose?arrow_forwardGiven the following: Cp of Br: 0.226 J/g-K BP of Br: 59 deg C density of Br: 3.12 g/mL Hvap of Br: 30,900 J/mol V of liquid Br solution: 50 mL Cp of Br2: 0.473 J/g-K What is the total heat required in heating Br from 25 to 75 deg C?arrow_forward
 Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press
Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON
Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education
Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY
Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning
Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY
Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY





