
In 1976, a team of chemists in the United Kingdom was developing new insecticides by modifying sugars with chlorine (Cl2), phosgene (Cl2CO), and other toxic gases. One young member of the team misunderstood his verbal instructions to “test” a new molecule. He thought he had been told to “taste” it. Luckily for him, the molecule was not toxic, but it was very sweet. It became the food additive sucralose.
Sucralose has three chlorine atoms substituted for three hydroxyl groups of sucrose (table sugar):
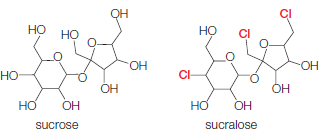
The altered sugar binds so strongly to the sweet-taste receptors on the tongue that the human brain perceives it as 600 times sweeter than sucrose. Sucralose was originally marketed as an artificial sweetener called Splenda®, but it is now available under several other brand names.
Researchers investigated whether the body recognizes sucralose as a carbohydrate by feeding sucralose labeled with 14C to volunteers. Analysis of the radioactive molecules in the volunteers’ urine and feces showed that 92.8 percent of the sucralose passed through the body without being altered.
Some people are worried that the chlorine atoms impart toxicity to sucralose. How would you respond to that concern?
Trending nowThis is a popular solution!

Chapter 3 Solutions
Biology: The Unity and Diversity of Life (MindTap Course List)
Additional Science Textbook Solutions
Loose Leaf For Integrated Principles Of Zoology
Campbell Biology (10th Edition)
Human Physiology
Campbell Biology: Concepts & Connections (8th Edition)
Laboratory Experiments in Microbiology (11th Edition)
- Select the term in column B which best matches the description in column A. There are more terms then needed. Column A Column B 1. The pentose sugar found in DNA. a) Cis fatty acid b) Trans fatty acid c) Coenzyme d) Cofacipr 2. Commonly referred to as "insoluble fiber". 3. Considered to be an amphipathic molecule. 4. A molecule with the formula C18H3602 is probably a e) Phospholipid 5. Bond created during the formation of the primary structure of a protein. ) Glycogen 6. A non-protein organic molecule needed for proper enzyme functioning. g) Cellulose 7. Pyrimidine base found only in RNA. h) Galactose i) Fructose 8. Increasing the number of these molecules in the cell membrane would increase the permeability of the cell membrane. i) Thymine k) Uracil 9. Bond which connects nucleotide monomers together. 1) Fatty acid m) Disaccharide n) Peptide bond o) A-Helix p) B-Pleated Sheet q) Ribose r) Deoxyribose 10.arrow_forwardOne of the main sources of sphingosine in the body is in the cell membrane. What complication could arise from the biological synthesis of ceramide?arrow_forwardThe crude caffeine isolated from tea often has a green tinge. Why?arrow_forward
- All of the following are functions of nucleotides EXCEPT: (A) They form part of the structure of important redox carriers.(B) They are important metabolites that serve as secondary messengers. (C) They are polymerized to form DNA and RNA.(D) They are used as reductants during the recycling of reduced glutathione. (E) They function as high energy equivalents and energetic currency.arrow_forwardHow do you synthesize Chloramphenicol and show the mechanism? Explain step by step please. (If you can 1 sentence is enough.) (Drug Chemistry)arrow_forwardA toxin is glycopeptide, X that is composed of an octapeptide and a saccharide Y. Glycopeptide X is composed of a glycan Y and an octapeptide. The octapeptide was cleaved by trypsin, giving 2 tetrapeptides of exactly the same composition except for its N-terminals. When the octapeptide was cleaved by chymotrypsin, it gave a pentapeptide and a tripeptide. DNFB treatment gave DNP-ser. The composition of the octapeptide is lys, ser, trp.and leu. The leu:ser ratio is 4:1. 1. What is the sequence of this octapeptide? Saccharide Y is a tetrasaccharide. To determine its linkages, a person methylated glycopeptide X effectively methylating saccharide Y. Cleavage gave the peptide and the following products: 2,3,4-tri-O-methyl-a-D-glucopyranoside acid, 2,3-di-O-methyl-ß-D- glucopyranoside, 1,3,4-tri-O- methyl-ß-D-glucuronic acid treatments to the isolated tetrasaccharide gave the following results: and 1,4,6-tri-O-methyl-ß-D-N- acetylgalactosamine. Further 1. A. Treatment of saccharide Y with an…arrow_forward
- Put the following anions in order from most nucleophillic to least nucleophillic: Br- I- F- Cl-arrow_forwardBlueberries have proanthocyanidins, which contain catechin monomer, dimer, and trimer, and polymers of flavan-3-ol. Proanthocyanidins have bacterial anti-sticking properties that create a thin wall of protection on tissues. This anti-sticking property helps prevent pathogens that act against the stomach wall and tooth enamel. There are also antioxidant properties that are more effective than vitamin E and C. Blueberries also contain manganese which is a crucial mineral for human functioning. Manganese is a component of the bone. An intake of manganese helps structure and strengthen the human bones. provide at least one alternative food item that provides one or more of the same macromolecules and/or minerals. Is your alternative healthier than the original selection? Are there any associated physiological disorders if one or more of the minerals provided were lacking from an individual’s diet?arrow_forwardThe death cap mushroom (Amanita phalloides) is considered one of the world’s deadliest mushrooms. The toxicity of the Amanita species is due to the presence of two groups of toxins known as amatoxins and phallotoxins, both made of multiple cyclic peptide chains (circular sequence of bonds). Do some research about the death cap and answer: What makes the Amanita phalloides toxins so harmful that even one cap can kill an adult?arrow_forward
- A decapeptide which could resist burns was isolated from an endemic plant from Mt. Mayon. Deduce the amino acid sequence of the decapeptide given the following informations. Mercaptoethanol gave two pentapeptides F1 and F2. Chymotrypsin treatment of F1 releases S and a tetrapeptide (C,K,F,M) while F2 gave a dipeptide (M,S) and a tripeptide (C,F,K) F1 when treated with CNBr released a free homoserine lactone and a tetrapeptide. Trypsin treatment of F1 gave a tripeptide and a dipeptide. F2 treated with carboxypeptidase released free M while with trypsin gave free K and a tetrapeptide.arrow_forwardDefine the following: 1. Cofactor 2. Coenzyme 3. "-ase" endingarrow_forwardCottage cheese and many other cheeses are produced by adding the enzyme rennin to milk, producing curds and whey. a) Design an experiment to determine the pH at which rennin is most effective. How will you measure the results? b) What are tests that can be used to detect protein, sugars, and fats? How can these tests to determine the chemical makeup of curds and whey?arrow_forward
