
Organic Chemistry
8th Edition
ISBN: 9781305580350
Author: William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 29.6, Problem FQ
Interpretation Introduction
Interpretation:
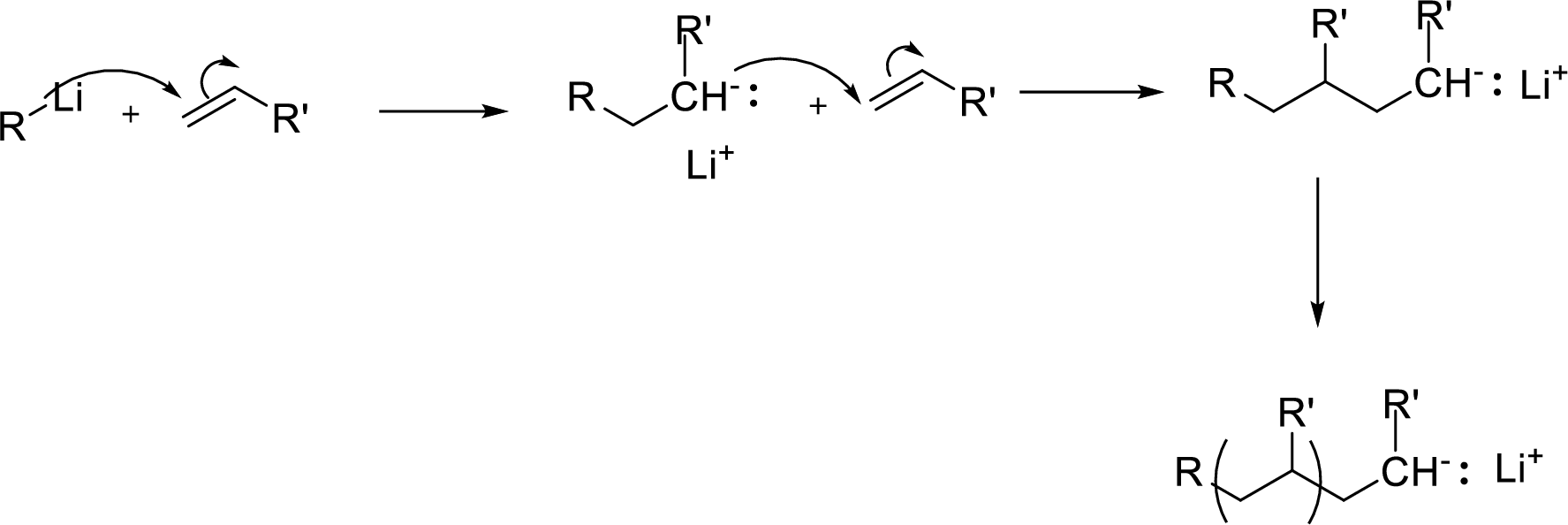
Among the given conditions, the condition that lowers the length of the
Concept Introduction:
Anionic
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
POLYMER CHEMISTRY
Show the mechanism of each item and their products.
For an addition reaction, why does free energy, ΔG, becomes more negative with decreasing temperature?
a. The negative enthalpy dominates at low temperature.
b. The negative entropy dominates at low temperature.
c. The positive entropy dominates at low temperature.
d. The positive enthalpy dominates at low temperature.
Reaction mechanism.
Chapter 29 Solutions
Organic Chemistry
Ch. 29.2 - Prob. 29.1PCh. 29.5 - Prob. 29.2PCh. 29.6 - Prob. AQCh. 29.6 - Prob. BQCh. 29.6 - Prob. CQCh. 29.6 - Prob. DQCh. 29.6 - Prob. EQCh. 29.6 - Prob. FQCh. 29.6 - Prob. 29.3PCh. 29.6 - Prob. 29.4P
Ch. 29 - Prob. 29.5PCh. 29 - Prob. 29.6PCh. 29 - Prob. 29.7PCh. 29 - Prob. 29.8PCh. 29 - Prob. 29.9PCh. 29 - Prob. 29.10PCh. 29 - Prob. 29.11PCh. 29 - Prob. 29.12PCh. 29 - Prob. 29.13PCh. 29 - Prob. 29.14PCh. 29 - Prob. 29.15PCh. 29 - Prob. 29.16PCh. 29 - Polycarbonates (Section 29.5C) are also formed by...Ch. 29 - Prob. 29.18PCh. 29 - Prob. 29.19PCh. 29 - Prob. 29.20PCh. 29 - Prob. 29.21PCh. 29 - Draw a structural formula of the polymer resulting...Ch. 29 - Prob. 29.23PCh. 29 - Prob. 29.24PCh. 29 - Prob. 29.25PCh. 29 - Select the monomer in each pair that is more...Ch. 29 - Prob. 29.27PCh. 29 - Prob. 29.28PCh. 29 - Prob. 29.29PCh. 29 - Prob. 29.30PCh. 29 - Prob. 29.31PCh. 29 - Prob. 29.32PCh. 29 - Prob. 29.33PCh. 29 - Radical polymerization of styrene gives a linear...Ch. 29 - Prob. 29.35PCh. 29 - Prob. 29.36PCh. 29 - Prob. 29.37PCh. 29 - Prob. 29.38P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Is the rate of the cleaving reaction relatively fast or relatively slow? Explain your reasoning.arrow_forwardWhich reaction occurs more quicklyarrow_forwardA carbocation is an intermediate formed in certain organic reactions. Chemically, what can happen to a carbocation? A carbocation can Select one: O A....bond to a nucleophile. B. ...lose a beta hydrogen ion to form a pi bond. O C. ...rearrange to a more stable carbocation. O D. All of the abovearrow_forward
- Consider the energy diagram in the Figure below. Which one of the following statements is *not* correct? reagents products Progress of the reaction A. The energy diagram represents information for a mechanism with three elementary steps. B. The energy diagram shows that the first elementary step is the slowest. C. The energy diagram represents an exothermic reaction. D. The energy diagram shows that the reaction has three transition states and two reactive intermediates. Energyarrow_forwardusing curly arrows step by step write the reaction mechanismarrow_forwardFor the following clauses reactions, draw the correct organic productsarrow_forward
- What is the rearrangement?arrow_forwardFruits are sometimes picked while green and transported long distances, after which they are exposed to ethene gas, which speeds the ripening process. Research and write a short paper detailing the advantages and disadvantages of using ethene to speed the ripening of fruits.arrow_forwardPressure affects the rate of reaction. TRUE or FALSEarrow_forward
- In this reaction, a tetrahedral intermediate is formed. What is the tetrahedral intermediate called and what does this signify about the molecule? The intermediate is known as the zwitterionic tetrahedral intermediate. A 1) zwitterion possesses an overall charge of 1. Separate functional groups are positively and negatively charged and the sum of these functional groups is one. The intermediate is known as the transition state. A transition state has no 2) overall electric charge. Separate functional groups are positively and negatively charged and the sum of these functional groups is zero. The intermediate is known as the transition state. A transition state has an 3) overall electric charge of 1. Separate functional groups are positively and negatively charged and the sum of these functional groups is one. The intermediate is known as the zwitterionic tetrahedral intermediate. A 4) zwitterion possesses no overall electrical charge, but it contains separate functional groups that are…arrow_forwardPLEASE COMPLETE THE REACTION. IT IS LIKE THIS IN BOOK .arrow_forwardChemistry Draw the main product and if the reaction can't proceed explain whyarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Chemistry for Engineering StudentsChemistryISBN:9781285199023Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781285199023Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:9781285199023
Author:Lawrence S. Brown, Tom Holme
Publisher:Cengage Learning