
Organic Chemistry
5th Edition
ISBN: 9780078021558
Author: Janice Gorzynski Smith Dr.
Publisher: McGraw-Hill Education
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 25, Problem 25.44P
Rank the nitrogen atoms in each compound in order of increasing basicity. Isoniazid is a drug used to treat tuberculosis, whereas histamine (Section 25.6B) causes the runny nose and watery eyes associated with allergies.
a. 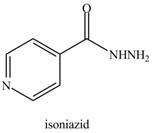 b.
b. 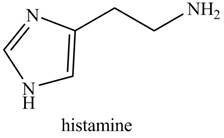
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Rank the nitrogen atoms in each compound in order of increasing basicity. Isoniazid is a drug used to treat tuberculosis, whereas histamine (Section 25.6B) causes the runny nose and watery eyes associated with allergies.
Rank the nitrogen atoms in each compound in order of increasing basicity. Isoniazid is a drug used to treat tuberculosis,
whereas histamine (Section 25.6B) causes the runny nose and watery eyes associated with allergies.
NH2
NHNH2
b.
а.
isoniazid
histamine
What carbonyl compound and amine are formed by the hydrolysis of each compound?
Chapter 25 Solutions
Organic Chemistry
Ch. 25 - Prob. 25.1PCh. 25 - Prob. 25.2PCh. 25 - Prob. 25.3PCh. 25 - Prob. 25.4PCh. 25 - Prob. 25.5PCh. 25 - Prob. 25.6PCh. 25 - Problem 25.7
Draw the product of each...Ch. 25 - Prob. 25.8PCh. 25 - Prob. 25.9PCh. 25 - Prob. 25.10P
Ch. 25 - Prob. 25.11PCh. 25 - Prob. 25.12PCh. 25 - Prob. 25.13PCh. 25 - Prob. 25.14PCh. 25 - Prob. 25.15PCh. 25 - Prob. 25.16PCh. 25 - Prob. 25.17PCh. 25 - Problem 25.18
Write out steps to show how each of...Ch. 25 - Prob. 25.19PCh. 25 - Prob. 25.20PCh. 25 - Prob. 25.21PCh. 25 - Problem 25.22
Which nitrogen atom in each compound...Ch. 25 - Prob. 25.23PCh. 25 - Prob. 25.24PCh. 25 - Prob. 25.25PCh. 25 - Prob. 25.26PCh. 25 - Prob. 25.27PCh. 25 - Problem 25.28
Draw the major product formed in...Ch. 25 - Prob. 25.29PCh. 25 - Prob. 25.30PCh. 25 - Problem 25.31
Devise a synthesis of each compound...Ch. 25 - Prob. 25.32PCh. 25 - Problem 25.33
What starting materials are needed...Ch. 25 - Problem 25.34
(a) What two components are needed...Ch. 25 - Prob. 25.35PCh. 25 - Prob. 25.36PCh. 25 - 25.37 Varenicline (trade name Chantix) is a drug...Ch. 25 - Give a systematic or common name for each...Ch. 25 - Prob. 25.39PCh. 25 - 25.40 How many stereogenic centers are present in...Ch. 25 - 25.41 Rank the compounds in each group in order of...Ch. 25 - 25.42 Decide which atom in each molecule is most...Ch. 25 - 25.43 Explain why pyrimidine is less basic than...Ch. 25 - 25.44 Rank the nitrogen atoms in each compound in...Ch. 25 - 25.45 Explain why nitroaniline is a stronger base...Ch. 25 - 25.46 Explain the observed difference in the...Ch. 25 - 25.47 Why is pyrrole more acidic than...Ch. 25 - Prob. 25.48PCh. 25 - Prob. 25.49PCh. 25 - Prob. 25.50PCh. 25 - 25.51 How would you separate toluene , benzoic...Ch. 25 - 25.52 Draw the products formed when methylaniline ...Ch. 25 - Prob. 25.53PCh. 25 - Prob. 25.54PCh. 25 - 25.55 Draw the organic products formed in each...Ch. 25 - Prob. 25.56PCh. 25 - 25.57 Identify A, B, and C, three intermediates in...Ch. 25 - Prob. 25.58PCh. 25 - Prob. 25.59PCh. 25 - 25.60 A chiral amine A having the configuration...Ch. 25 - 25.61 Draw a stepwise mechanism for each...Ch. 25 - 25.62 Draw a stepwise mechanism for the following...Ch. 25 - Prob. 25.63PCh. 25 - 25.64 Tertiary aromatic amines react with and ...Ch. 25 - 25.65 Devise a synthesis of each compound from...Ch. 25 - Prob. 25.66PCh. 25 - Prob. 25.67PCh. 25 - Prob. 25.68PCh. 25 - Prob. 25.69PCh. 25 - 25.70 Devise a synthesis of each biologically...Ch. 25 - 25.71 Devise a synthesis of each compound from...Ch. 25 - 25.72 Three isomeric compounds A, B, and C, all...Ch. 25 - 25.73 Treatment of compound D with LiAlH4 followed...Ch. 25 - Prob. 25.74PCh. 25 - 25.75 Rank the following compounds in order of...Ch. 25 - Prob. 25.76PCh. 25 - Prob. 25.77PCh. 25 - Prob. 25.78P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Rank each set of compounds in order of increasing basicity.aniline, pyrrole, pyridine, piperidinearrow_forwardWhat carbonyl compound and amine or alcohol are needed to prepare each product?arrow_forward17. Rank the acid derivatives with respect to their reactivity with water. acid halide > ester > acid anhydride > amide acid anhydride > amide > acid halide > ester amide > ester > acid anhydride > acid halide acid halide > acid anhydride > ester > amide A) B) C) D) 18. Disregarding A) 3 stereoisomers, how many different enols can the B-diketone CH3COCH₂COCH₂CH3 form? B) 1 C) 4 D) O E) 2arrow_forward
- 18.64 The following two compounds each exhibit two heteroatoms (one nitrogen atom and one oxygen atom). :0 Compound A -N Compound B O In compound A, the lone pair on the nitrogen atom is more likely to function as a base. However, in compound B, the lone pair on the oxygen atom is more likely to function as a base. Explain this difference.arrow_forwardRank the compounds in attached group in order of increasing acidity.arrow_forwardAnswer the following questions about atomoxetine, a drug used to treatattention deficit hyperactivity disorder (ADHD). a.) What amides can be reduced to form atomoxetine?b.) What starting materials can be used to form atomoxetine by reductiveamination? Draw all possible methods.c.) What products are formed by Hofmann elimination of atomoxetine?arrow_forward
- Decide which N atom in each molecule is most basic, and draw theproduct formed when each compound is treated with CH3CO2H.Zolpidem (trade name Ambien) is used to treat insomnia, whereasaripiprazole (trade name Abilify) is used to treat depression,schizophrenia, and bipolar disorders.arrow_forwardRank the labeled N atoms in the anticancer drug imatinib (trade nameGleevec) in order of increasing basicity. Imatinib, sold as a salt withmethanesulfonic acid (CH3SO3H), is used for the treatment of chronicmyeloid leukemia as well as certain gastrointestinal tumors.arrow_forwardDraw the product formed when phenylacetic acid (C6H5CH2COOH) is treated with each reagent. With some reagents, no reaction occurs. (A - F)arrow_forward
- How would you convert benzoic acid (C6H5CO2H) to each compound?arrow_forwardThe order of increasing acidity is Phenol < meta-cyanophenol < ortho-cyanophenol 1.how about para-cyanophenol??does it has higher acidicity than both ortho-cyanophenol and meta cyanophenol or lower?? 2.why does ortho-cyanophenol have higher acidicity than meta cyanophenol?? 3. why doesmeta cyanophenol have higher acidicity than Phenolarrow_forwardwhy pyrazole is more basic than pyridine?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning

Organic And Biological Chemistry
Chemistry
ISBN:9781305081079
Author:STOKER, H. Stephen (howard Stephen)
Publisher:Cengage Learning,

General, Organic, and Biological Chemistry
Chemistry
ISBN:9781285853918
Author:H. Stephen Stoker
Publisher:Cengage Learning
General Chemistry | Acids & Bases; Author: Ninja Nerd;https://www.youtube.com/watch?v=AOr_5tbgfQ0;License: Standard YouTube License, CC-BY