
(a)
Interpretation: The structure corresponding to given compound is to be drawn.
Concept introduction:
1. The hydrocarbon is named after the longest carbon chain.
2. The parent hydrocarbon containing amino group is named as
3. When the nitrogen atom of amine is substituted with alkyl groups, then the amine is named with prefix
Answer to Problem 25.3P
The structure of the given compound is,
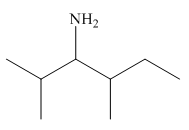
Explanation of Solution
The IUPAC name of the compound is

Figure 1
The structure of the given compound is shown in Figure 1.
(b)
Interpretation: The structure corresponding to given compound is to be drawn.
Concept introduction: IUPAC nomenclature is a systematic way of naming the organic compounds. The basic principles of IUPAC naming for hydrocarbon are:
1. The hydrocarbon is named after the longest carbon chain.
2. The parent hydrocarbon containing amino group is named as alkane with suffix amine.
3. When the nitrogen atom of amine is substituted with alkyl groups, then the amine is named with prefix
Answer to Problem 25.3P
The structure of the given compound is,

Explanation of Solution
The IUPAC name of the compound is

Figure 2
The structure of the given compound is shown in Figure 2.
(c)
Interpretation: The structure corresponding to given compound is to be drawn.
Concept introduction: IUPAC nomenclature is a systematic way of naming the organic compounds. The basic principles of IUPAC naming for hydrocarbon are:
1. The hydrocarbon is named after the longest carbon chain.
2. The parent hydrocarbon containing amino group is named as alkane with suffix amine.
3. When the nitrogen atom of amine is substituted with alkyl groups, then the amine is named with prefix
Answer to Problem 25.3P
The structure of the given compound is,
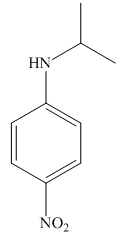
Explanation of Solution
The IUPAC name of the compound is

Figure 3
The structure of the given compound is shown in Figure 3.
(d)
Interpretation: The structure corresponding to given compound is to be drawn.
Concept introduction: IUPAC nomenclature is a systematic way of naming the organic compounds. The basic principles of IUPAC naming for hydrocarbon are:
1. The hydrocarbon is named after the longest carbon chain.
2. The parent hydrocarbon containing amino group is named as alkane with suffix amine.
3. When the nitrogen atom of amine is substituted with alkyl groups, then the amine is named with prefix
Answer to Problem 25.3P
The structure of the given compound is,
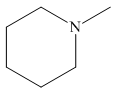
Explanation of Solution
The IUPAC name of the compound is

Figure 4
The structure of the given compound is shown in Figure 4.
(e)
Interpretation: The structure corresponding to given compound is to be drawn.
Concept introduction: IUPAC nomenclature is a systematic way of naming the organic compounds. The basic principles of IUPAC naming for hydrocarbon are:
1. The hydrocarbon is named after the longest carbon chain.
2. The parent hydrocarbon containing amino group is named as alkane with suffix amine.
3. When the nitrogen atom of amine is substituted with alkyl groups, then the amine is named with prefix
Answer to Problem 25.3P
The structure of the given compound is,
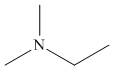
Explanation of Solution
The IUPAC name of the compound is

Figure 5
The structure of the given compound is shown in Figure 5.
(f)
Interpretation: The structure corresponding to given compound is to be drawn.
Concept introduction: IUPAC nomenclature is a systematic way of naming the organic compounds. The basic principles of IUPAC naming for hydrocarbon are:
1. The hydrocarbon is named after the longest carbon chain.
2. The parent hydrocarbon containing amino group is named as alkane with suffix amine.
3. When the nitrogen atom of amine is substituted with alkyl groups, then the amine is named with prefix
Answer to Problem 25.3P
The structure of the given compound is,
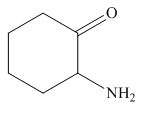
Explanation of Solution
The IUPAC name of the compound is

Figure 6
The structure of the given compound is shown in Figure 6.
(g)
Interpretation: The structure corresponding to given compound is to be drawn.
Concept introduction: IUPAC nomenclature is a systematic way of naming the organic compounds. The basic principles of IUPAC naming for hydrocarbon are:
1. The hydrocarbon is named after the longest carbon chain.
2. The parent hydrocarbon containing amino group is named as alkane with suffix amine.
3. When the nitrogen atom of amine is substituted with alkyl groups, then the amine is named with prefix
Answer to Problem 25.3P
The structure of the given compound is,
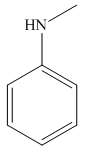
Explanation of Solution
The IUPAC name of the compound is

Figure 7
The structure of the given compound is shown in Figure 7.
(h)
Interpretation: The structure corresponding to given compound is to be drawn.
Concept introduction: IUPAC nomenclature is a systematic way of naming the organic compounds. The basic principles of IUPAC naming for hydrocarbon are:
1. The hydrocarbon is named after the longest carbon chain.
1. The parent hydrocarbon containing amino group is named as alkane with suffix amine.
2. When the nitrogen atom of amine is substituted with alkyl groups, then the amine is named with prefix
Answer to Problem 25.3P
The structure of the given compound is,
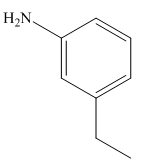
Explanation of Solution
The IUPAC name of the compound is

Figure 8
The structure of the given compound is shown in Figure 8.
Want to see more full solutions like this?
Chapter 25 Solutions
Organic Chemistry
- Draw condensed and skeletal structures for each of the following amines a. 2-methyl-N-propyl-1-propanamine b. N-ethylethanamine c. 5-methyl-1-hexanamine d. methyldipropylaminee. e. N,N-dimethyl-3-pentanamine f. cyclohexylethylmethylaminearrow_forwardErythronolide B is the biological precursor of erythromycin, a broad-spectrum antibiotic. What functional group Erythronolide B does contain? a. b. H₂CH₂C C. H₂C 1 H₂C 2 3 4 O Amide d. Amine OH Erythronolide B Ketone Aldehyde a CH₂ b C d CH₂ OH JCH₂ 'OH OHarrow_forwardWhat are the functional groups present in this antibacterial antibiotic? A. Amide, thioether, aldehyde, phenol, carboxylic acid B. Amide, thioether, ketone, amine, phenol, carboxylic acid C. Amide, thioether, ketone, phenol, carboxylic acid D. Thioether, ketone, amine, phenol, carboxylic acid A brief explanation would be highly appreciated + upvotearrow_forward
- explain the solubility behavior of each representatives amine in water a. aniline b. diethylamine c. N,N-diethylanilinearrow_forwardH. What type of amine is the functional group above (The R is a carbon group) R a. primary Ob. tertiary Oc. secondaryarrow_forward2. Draw out the following compounds. a. N-methylaniline b. Triisopropylamine c. N,N-dipropylhexylamine d. 1,5-pentanediamine (Also known as Cadaverine for its smell)arrow_forward
- The hydrolysis of an amide in acidic conditions forms A. a carboxylate salt and an alcohol B. a carboxylate salt and an amine C. an alcohol and an amine salt (an ammonium ion) D. a carboxylic acid and an amine salt (an ammonium ion)arrow_forwardAmide hydrolysis in basic conditions forms A. a carboxylic acid and an amine B. a carboxylate salt and an amine 3. an ester and an amine 4. a carboxylic acid and an amine saltarrow_forwardWhat are the functional groups present in this antibacterial antibiotic? A. Nitro, phenyl, amine, carbonyl, hydroxyl B. Nitro, phenyl, amine, carbonyl, C. Nitro, phenyl, amine, hydroxyl D. Nitro, phenol, amine, carbonyl, hydroxyl A brief explanation would be highly appreciated + upvotearrow_forward
- Which amine below is an primary aromatic amine? a. N-methyl-2-phenylethanamine b. All of the given c. 3-methylbenzamine d. N-ethylbenzaminearrow_forward1. Draw the structure for each compound and classify the amine as primary, secondary, or tertiary. a. dimethylamine b. diethylmethylamine c. 2-aminoethanolarrow_forwardC. Amides 1. Amidation a. Acetic acid + ammoniaarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





