
Concept explainers
(a)
Interpretation:
Whether crotonate, a C4 species is a (1) hydroxy acid, (2) keto acid, (3) saturated acid, or (4) unsaturated acid has to be identified.
Concept introduction:
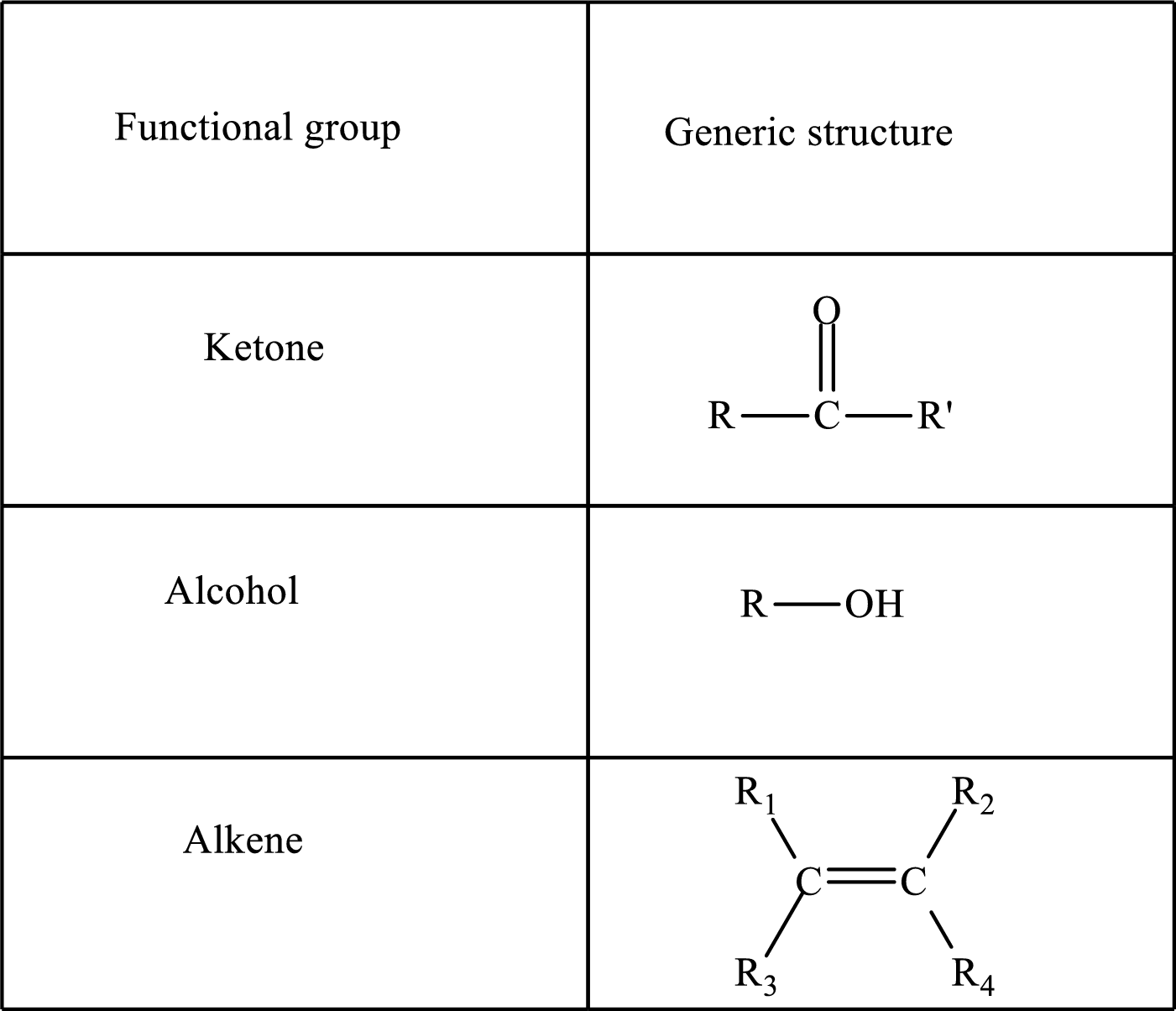
Here, R and R’ represent an alkyl group. In alkene, R1, R2, R3, and R4 can be the same or different or can be hydrogen.
Keto acid has a
A carboxylate group is formed by the removal of the acidic hydrogen from the carboxylic group. The conjugate base is formed by the removal of acidic hydrogen from the corresponding acid.
(b)
Interpretation:
Whether oxaloacetate, a C4 species is a (1) hydroxy acid, (2) keto acid, (3) saturated acid, or (4) unsaturated acid has to be identified.
Concept introduction:
Functional groups are defined as the group of atoms which are attached to the carbon backbone of organic compounds. These are generally heteroatoms which are attached to the parent hydrocarbon chain. Some examples of functional groups are as follows:
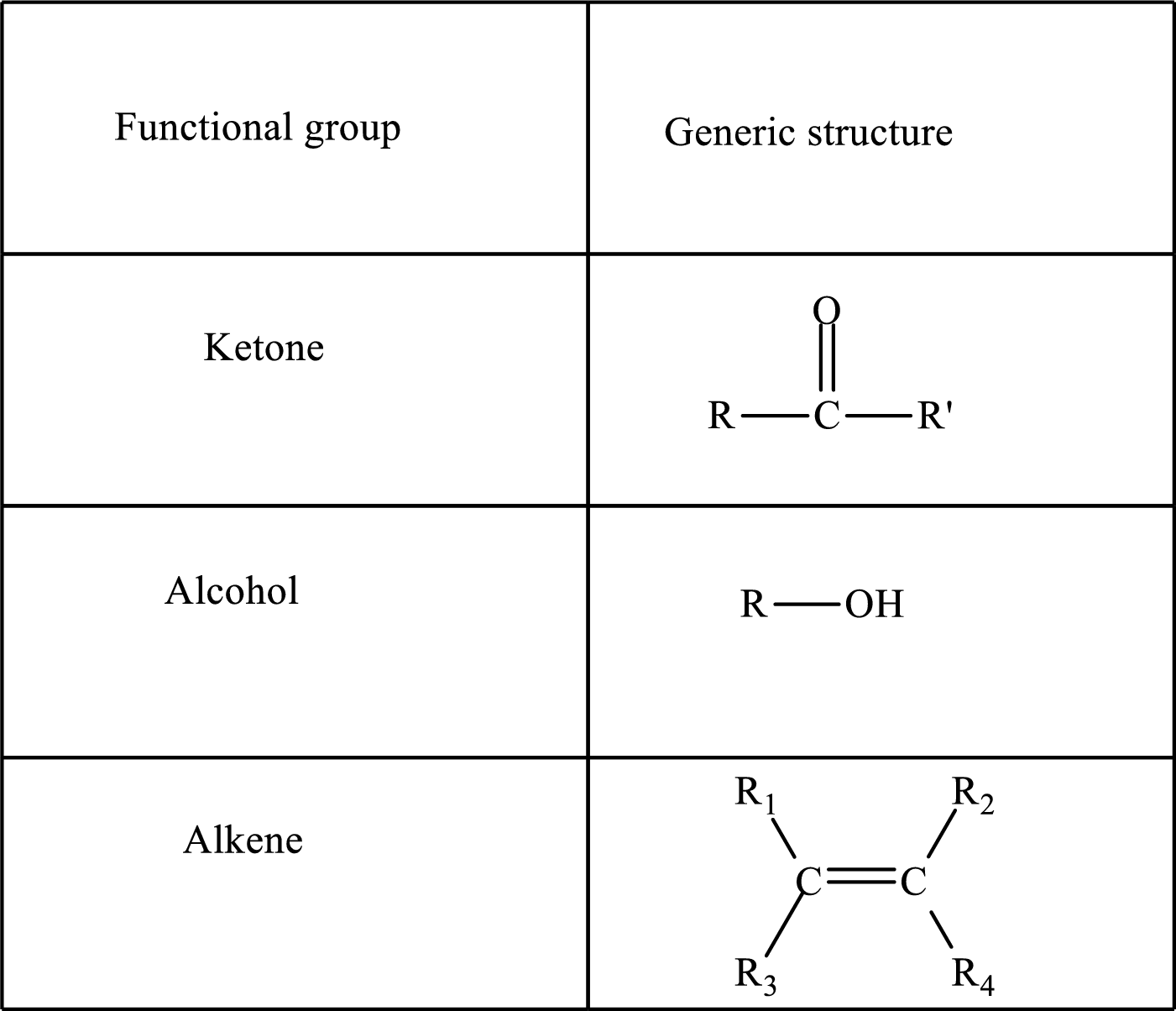
Here, R and R’ represent an alkyl group. In alkene, R1, R2, R3, and R4 can be the same or different or can be hydrogen. Alkanes are saturated hydrocarbons that contain covalently bonded hydrogen and carbon atoms. Alkenes have a double bond, hence; they are unsaturated compounds.
Keto acid has a ketone and a carboxylic acid (-COOH) group. Hydroxy acid has a hydroxy (-OH) group and a carboxylic acid (-COOH) group. Saturated acids contain single bonds between carbon atoms and a carboxylic group. Unsaturated acid contains a double or triple bond between carbon atoms and a carboxylic group.
A carboxylate group is formed by the removal of the acidic hydrogen from the carboxylic group. The conjugate base is formed by the removal of acidic hydrogen from the corresponding acid.
(c)
Interpretation:
Whether acetoacetate, a C4 species is a (1) hydroxy acid, (2) keto acid, (3) saturated acid, or (4) unsaturated acid has to be identified.
Concept introduction:
Functional groups are defined as the group of atoms which are attached to the carbon backbone of organic compounds. These are generally heteroatoms which are attached to the parent hydrocarbon chain. Some examples of functional groups are as follows:
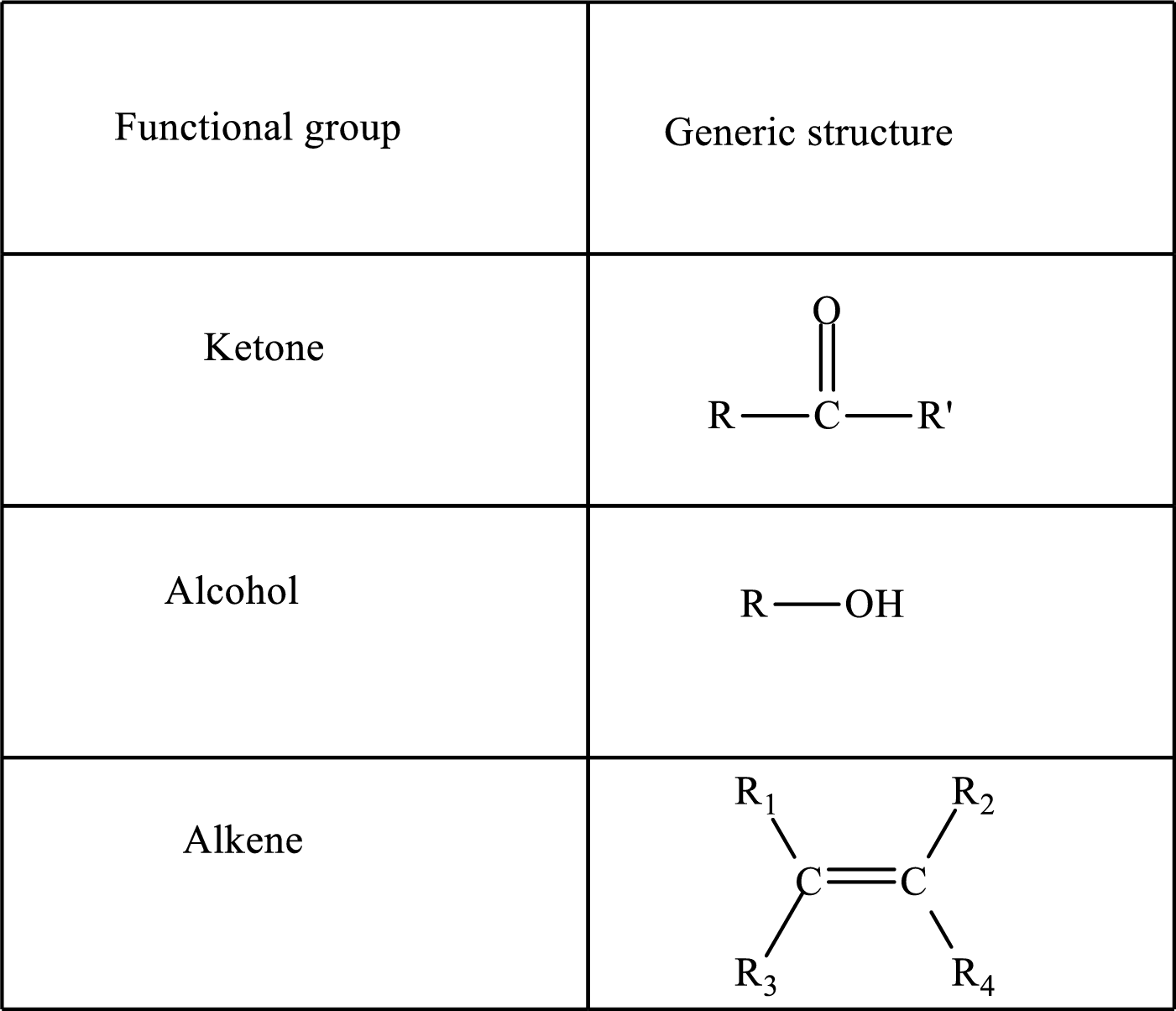
Here, R and R’ represent an alkyl group. In alkene, R1, R2, R3, and R4 can be the same or different or can be hydrogen. Alkanes are saturated hydrocarbons that contain covalently bonded hydrogen and carbon atoms. Alkenes have a double bond, hence; they are unsaturated compounds.
Keto acid has a ketone and a carboxylic acid (-COOH) group. Hydroxy acid has a hydroxy (-OH) group and a carboxylic acid (-COOH) group. Saturated acids contain single bonds between carbon atoms and a carboxylic group. Unsaturated acid contains a double or triple bond between carbon atoms and a carboxylic group.
A carboxylate group is formed by the removal of the acidic hydrogen from the carboxylic group. The conjugate base is formed by the removal of acidic hydrogen from the corresponding acid.
(d)
Interpretation:
Whether malate, a C4 species is a (1) hydroxy acid, (2) keto acid, (3) saturated acid, or (4) unsaturated acid has to be identified.
Concept introduction:
Functional groups are defined as the group of atoms which are attached to the carbon backbone of organic compounds. These are generally heteroatoms which are attached to the parent hydrocarbon chain. Some examples of functional groups are as follows:
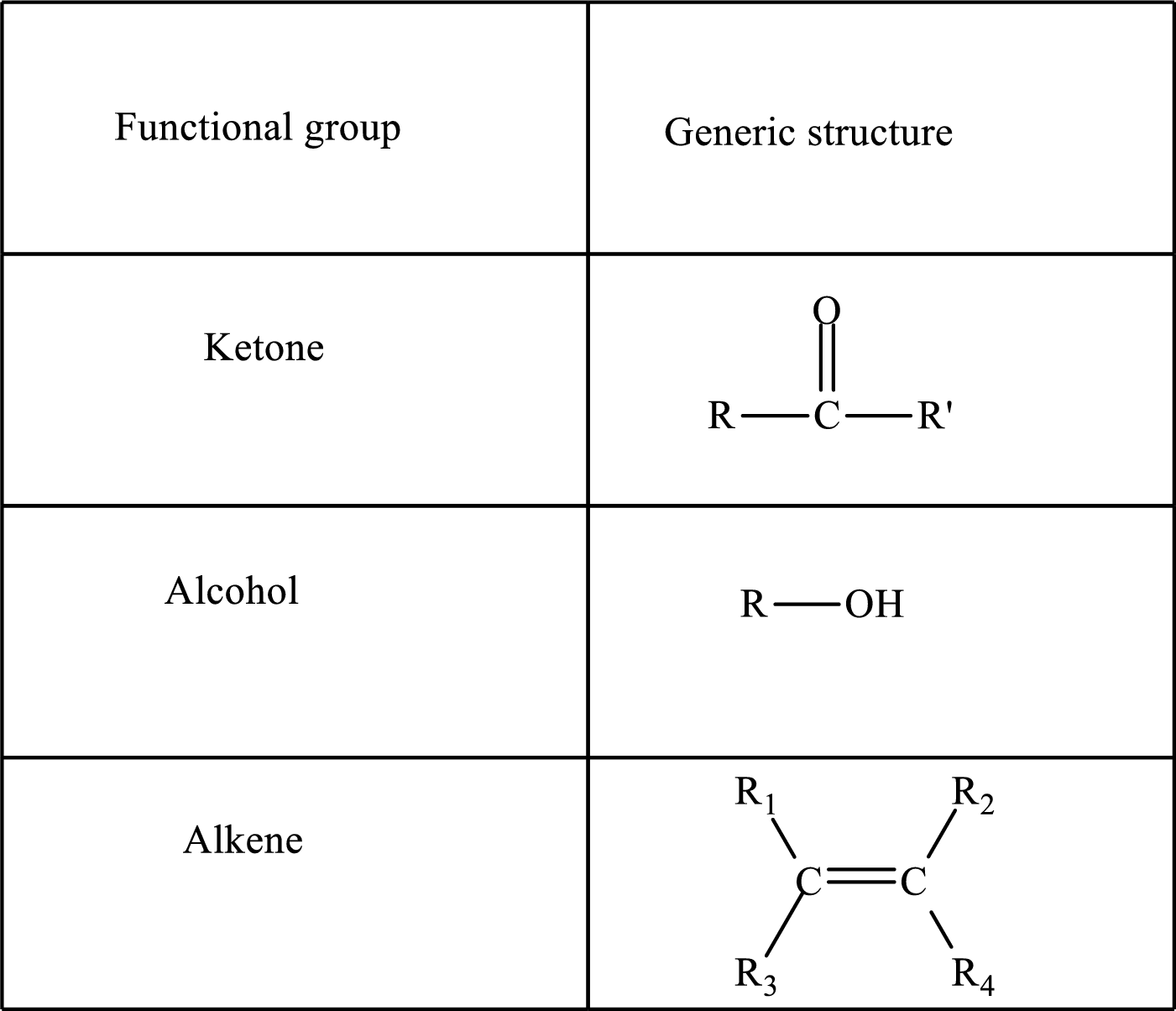
Here, R and R’ represent an alkyl group. In alkene, R1, R2, R3, and R4 can be the same or different or can be hydrogen. Alkanes are saturated hydrocarbons that contain covalently bonded hydrogen and carbon atoms. Alkenes have a double bond, hence; they are unsaturated compounds.
Keto acid has a ketone and a carboxylic acid (-COOH) group. Hydroxy acid has a hydroxy (-OH) group and a carboxylic acid (-COOH) group. Saturated acids contain single bonds between carbon atoms and a carboxylic group. Unsaturated acid contains a double or triple bond between carbon atoms and a carboxylic group.
A carboxylate group is formed by the removal of the acidic hydrogen from the carboxylic group. The conjugate base is formed by the removal of acidic hydrogen from the corresponding acid.
Want to see the full answer?
Check out a sample textbook solution
Chapter 25 Solutions
General, Organic, and Biological Chemistry
- What is a carbamoyl group?arrow_forwardRefer to the structure 1. The metabolic pathway is an example of a. Oxidative dealkylation b. Reduction c. Oxidative deamination d. Hydrolysis 2. This pathway is true for what kind of drugs a. Endogenous amides b. Endogenous amines c. Radical species d. Endogenous amino acidsarrow_forwarddescription for the term rancidity of oils two causes of rancidity of oils acid value of oilsarrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Chemistry In FocusChemistryISBN:9781305084476Author:Tro, Nivaldo J., Neu, Don.Publisher:Cengage Learning
Chemistry In FocusChemistryISBN:9781305084476Author:Tro, Nivaldo J., Neu, Don.Publisher:Cengage Learning- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
 General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,





