
Interpretation: The question is to describe Pauli’s exclusion principle and Hund’s rule.
Concept Introduction: Pauli's exclusion principle and Hund's rule are primarily employed in chemistry to clarify or ascertain the electron shell structure of atoms and to foretell which atoms are most likely to donate electrons.
Answer to Problem 102A
According to Hund's principle, electrons should first fill lower energy levels in the atomic orbitals before moving up to higher states.
No two electrons may have the same four quantum numbers, according to Pauli's Exclusion Principle.
Explanation of Solution
Given information:
Pauli’s exclusion principle and Hund’s rule.
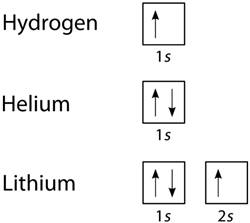
According to Pauli's Exclusion Principle, no two electrons in the same atom can have values for all four of their quantum numbers that are exactly the same.
In other words,
- Only two electrons may share an orbital.
- The spins of any two electrons sharing an orbital must be opposing.
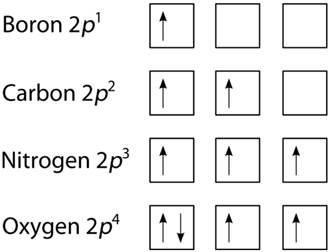
According to Hund's rule, all electrons in singly occupied orbitals have the same spin since every orbital in a subshell is singly occupied with one electron before any one orbital is doubly filled.
Hence, Pauli’s exclusion principle and Hund’s rule are stated above.
Chapter 25 Solutions
Chemistry 2012 Student Edition (hard Cover) Grade 11
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





