
Concept explainers
(a)
Interpretation:The structural formula of 3-methylpentanal needs to be drawn.
Concept Introduction:
(a)
Answer to Problem 2RQ
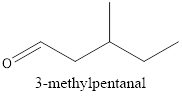
Explanation of Solution
The general formula for an aldehyde is R-CHO.
And the general formula for a ketone is R-COOR’.
Where R and R’ are alkyl group which may or may not be the same.
Therefore, the structural formula for 3-methylpentanal is
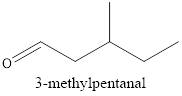
(b)
Interpretation:The structural formula of 3-methylpentanal needs to be drawn.
Concept Introduction:Ketone: A hydrocarbon derivativecontaining the carbonyl group bonded to twocarbon atoms.
Aldehyde: A hydrocarbon derivativecontaining the carbonylgroup at the end of thehydrocarbon chain.
(b)
Answer to Problem 2RQ

Explanation of Solution
The general formula for an aldehyde is RCHO.
And, the general formula for a ketone is RCOOR’.
Where R and R’ are alkyl group which may or may not be the same.
Therefore, the structural formula for 3-methyl-2-pentanone is

(c)
Interpretation:Thestructural formula of methyl phenyl ketone needs to be drawn.
Concept Introduction:Ketone: A hydrocarbon derivativecontaining the carbonyl group bonded to twocarbon atoms.
Aldehyde: A hydrocarbon derivativecontaining the carbonylgroup at the end of thehydrocarbon chain.
(c)
Answer to Problem 2RQ
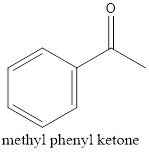
Explanation of Solution
The general formula for an aldehyde is RCHO.
And the general formula for a ketone is RCOOR’.
Where R and R’ are alkyl group which may or may not be the same.
Therefore, the structural formula for methyl phenyl ketone.
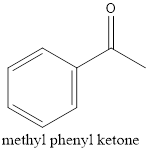
(d)
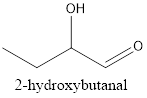
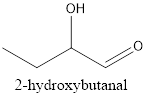
Interpretation:The structural formula of 2-hydroxybutanal needs to be drawn.
Concept Introduction:Ketone: A hydrocarbon derivativecontaining the carbonyl group bonded to twocarbon atoms.
Aldehyde: A hydrocarbon derivativecontaining the carbonylgroup at the end of thehydrocarbon chain.
(d)
Answer to Problem 2RQ
Explanation of Solution
The general formula for an aldehyde is RCHO.
And the general formula for a ketone is RCOOR’.
Where R and R’ are alkyl groupsthat may or may not be the same.
Therefore, the structural formula for 2-hydroxybutanal is
(E)
Interpretation:The structural formula of propanol needs to be determined.
Concept Introduction:Ketone: Ketone: A hydrocarbon derivativecontaining the carbonyl group bonded to twocarbon atoms.
Aldehyde: A hydrocarbon derivativecontaining the carbonylgroup at the end of thehydrocarbon chain.
(E)
Answer to Problem 2RQ

Explanation of Solution
The general formula for an aldehyde is RCHO.
And the general formula for a ketone is RCOOR’.
Where R and R’ are alkyl groups which may or may not be the same.
Therefore, the structural formula for propanol is

Chapter 20 Solutions
World of Chemistry
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





