
(a)
Interpretation:
The name of the given alkene has to be described.
Concept Introduction:
Alkenes can also be two types-
- Unbranched alkenes
- Branched alkenes
(a)
Answer to Problem 30A
The name of the above compound is 2-pentene.
Explanation of Solution
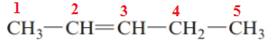
The principle chain contains five carbon atoms and one double bond between C2 and C3. So, the name of the above compound is 2-pentene.
(b)
Interpretation:
The name of the given alkene has to be described.
Concept Introduction:
Alkenes are acyclic or cyclic unsaturated hydrocarbons containing at least one carbon-carbon double bond (C=C) and the general molecular formula CnH2n (where n = number of carbon atoms). Simplest alkene among all is ethylene C2H4 which contains one carbon-carbon double bond.
Alkenes can also be two types-
- Unbranched alkenes
- Branched alkenes
(b)
Answer to Problem 30A
The name of the above compound is 2-pentene.
Explanation of Solution
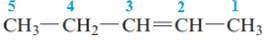
The principle chain contains five carbon atoms and one double bond between C2 and C3. So, the name of the above compound is 2-pentene.
(c)
Interpretation:
The name of the given alkene has to be described.
Concept Introduction:
Alkenes are acyclic or cyclic unsaturated hydrocarbons containing at least one carbon-carbon double bond (C=C) and the general molecular formula CnH2n (where n = number of carbon atoms). Simplest alkene among all is ethylene C2H4 which contains one carbon-carbon double bond.
Alkenes can also be two types-
- Unbranched alkenes
- Branched alkenes
(c)
Answer to Problem 30A
The name of the above compound is 2,5-dimethylhex-3-ene.
Explanation of Solution
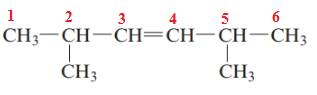
The principle chain contains six carbon atoms, two methyl substituents at C2 and C5 and one double bond between C3 and C4. So, the name of the above compound is 2,5-dimethylhex-3-ene.
(d)
Interpretation:
The name of the given alkene has to be described.
Concept Introduction:
Alkenes are acyclic or cyclic unsaturated hydrocarbons containing at least one carbon-carbon double bond (C=C) and the general molecular formula CnH2n (where n = number of carbon atoms). Simplest alkene among all is ethylene C2H4 which contains one carbon-carbon double bond.
Alkenes can also be two types-
- Unbranched alkenes
- Branched alkenes
(d)
Answer to Problem 30A
The name of the above compound is 3,4,5-trimethylhept-1-ene.
Explanation of Solution
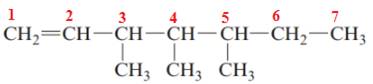
The principle chain contains seven carbon atoms, three methyl substituents at C3, C4 and C5 and one double bond between C1 and C2. So, the name of the above compound is 3,4,5-trimethylhept-1-ene.
Chapter 20 Solutions
World of Chemistry
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





