
Concept explainers
(a)
Interpretation:
The structural formula of the compound is to be written.
Concept introduction:
To write the structural formula,
(a)
Answer to Problem 51A

Explanation of Solution
Phenyl methyl
A ketone has general formula
It means the two alkyl groups of ketone are phenyl and methyl. So the structure is:

(b)
Interpretation:
The structural formula of the compound is to be written.
Concept introduction:
To write the structural formula, functional group, number of carbon atoms in the chain is to be identified.
(b)
Answer to Problem 51A
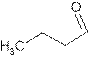
Explanation of Solution
The name butanal shows compound contains
Its general formula is
The word-root ‘but’ shows compound contains chain of four carbon atoms. So the structure is:
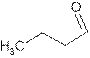
(c)
Interpretation:
The structural formula of the compound is to be written.
Concept introduction:
To write the structural formula, functional group, number of carbon atoms in the chain is to be identified.
(c)
Answer to Problem 51A

Explanation of Solution
The name of organic compound shows compound contains ketonic group.
A ketone has general formula
The name ‘but’ shows it has four carbon atoms.
So the structure is:

(d)
Interpretation:
The structural formula of the compound is to be written.
Concept introduction:
To write the structural formula, functional group, number of carbon atoms in the chain is to be identified.
(d)
Answer to Problem 51A
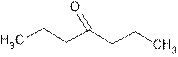
Explanation of Solution
Dipropyl ketone is common name of organic compound containing ketonic group.
A ketone has general formula
It means the two alkyl groups of ketone are propyl. So the structure is:
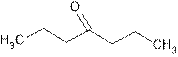
(e)
Interpretation:
The structural formula of the compound is to be written.
Concept introduction:
To write the structural formula, functional group, number of carbon atoms in the chain is to be identified.
(e)
Answer to Problem 51A
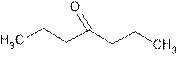
Explanation of Solution
The root name ‘heptan’ shows seven carbon atoms in the main chain which are single bonded.
The suffix ‘one’ shows it is a ketonic group. Its position is C-4.
Thus, the structure is
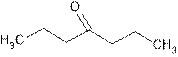
Chapter 20 Solutions
World of Chemistry
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





