
(a)
Interpretation:
The given chemical equation is to be completed.
Concept Introduction:
 C) and the general molecular formula of alkynes is CnH2n-2 (where n = number of carbon atoms). Simplest
C) and the general molecular formula of alkynes is CnH2n-2 (where n = number of carbon atoms). Simplest  CH)which contains one carbon-carbon triple bond.
CH)which contains one carbon-carbon triple bond.
(a)
Answer to Problem 29A
Explanation of Solution
The
(b)
Interpretation:
The given chemical equation is to be completed.
Concept Introduction:
Alkenes are acyclic or cyclic unsaturated hydrocarbons containing at least one carbon-carbon double bond (C=C) and the general molecular formula CnH2n (where n = number of carbon atoms). Simplest alkene among all is ethylene C2H4 which contains one carbon-carbon double bond.
(b)
Answer to Problem 29A
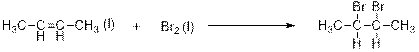
Explanation of Solution
This is an addition reaction between bromine and alkene. Saturated dibromo-
When 2-butene is treated with bromine, 2,3-dibomobutane is formed.
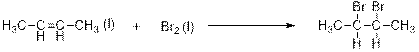
(c)
Interpretation:
The given chemical equation is to be completed.
Concept Introduction:
Alkynes are acyclic unsaturated hydrocarbons containing at least one carbon-carbon triple bond (C C) and the general molecular formula of alkynes is CnH2n-2 (where n = number of carbon atoms). Simplest alkene among all is acetylene C2H2 (HC
C) and the general molecular formula of alkynes is CnH2n-2 (where n = number of carbon atoms). Simplest alkene among all is acetylene C2H2 (HC CH)which contains one carbon-carbon triple bond.
CH)which contains one carbon-carbon triple bond.
(c)
Answer to Problem 29A

Explanation of Solution
When oxidation of alkyne is done, there occurs the formation of carbonyl compound. Terminal alkyne is oxidised to

Chapter 20 Solutions
World of Chemistry
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





