
Microbiology: An Introduction (13th Edition)
13th Edition
ISBN: 9780134605180
Author: Gerard J. Tortora, Berdell R. Funke, Christine L. Case, Derek Weber, Warner Bair
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
thumb_up100%
Chapter 2, Problem 8R
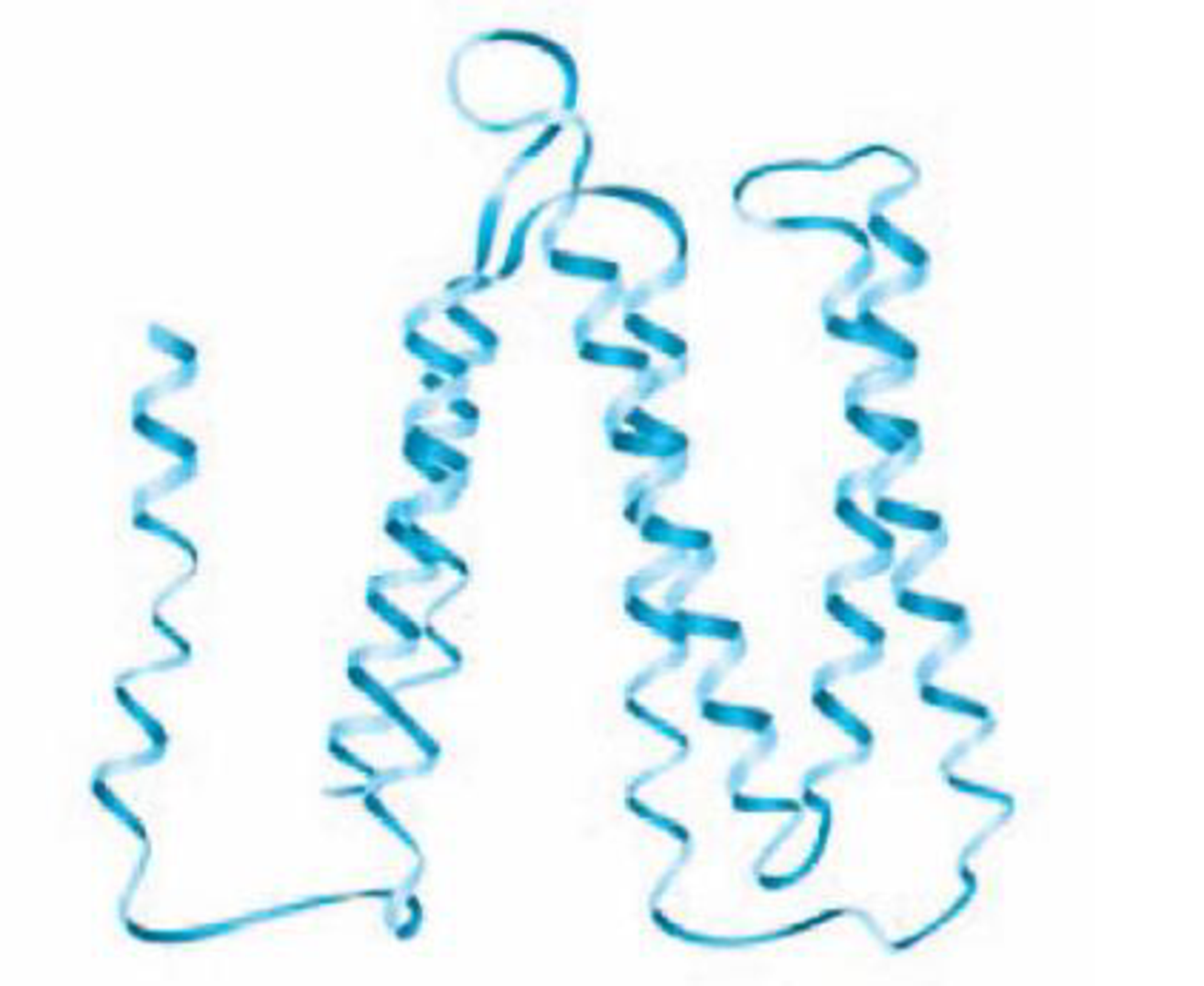
DRAW IT The following diagram shows the bacteriorhodopsin protein. Indicate the regions of primary, secondary, and tertiary structure. Does this protein have quaternary structure?
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Nearly all bacteria contain peptidoglycan. Review the structure of peptidoglycan by selecting the correct characteristics listed below.
Check All That Apply
Polymer of alternating N-acetylglucosamine (NAG) and N-acetylmuramic acid (NAM)
Contains a tetrapeptide that extends from N-acetylglucosamine (NAG)
Contains D-amino acids not found in proteins
All bacteria use a peptide interbridge to connect the sugar strands
Peptidoglycan is a strong and rigid structure
Which statement best describes the protein structure shown below?
This is an alpha helix.
This is a tertiary structure that consists almost entirely of beta strands.
This is a tertiary structure consisting of a roughly equal mixture of alpha helices and beta sheets.
This is a beta sheet.
This is a tertiary structure that consists almost entirely of alpha helices.
Draw a parallel beta sheet and an antiparallel beta sheet (three strands each). Show the
hydrogen bonding pattern as well as the positions of the R groups of the amino acids. You do not
need to draw the exact atoms; a simple representation is fine.
Chapter 2 Solutions
Microbiology: An Introduction (13th Edition)
Ch. 2 - What is a chemical element?Ch. 2 - DRAW IT Diagram the electronic configuration of a...Ch. 2 - What type of bond holds the following atoms...Ch. 2 - Classify the following types of chemical...Ch. 2 - Bacteria use the enzyme urease to obtain nitrogen...Ch. 2 - Classify the following as subunits of either a...Ch. 2 - DRAW IT The artificial sweetener aspartame, or...Ch. 2 - DRAW IT The following diagram shows the...Ch. 2 - Prob. 9RCh. 2 - Prob. 10R
Ch. 2 - Assume E. coli bacteria are grown in a nutrient...Ch. 2 - If Pseudomonas bacteria are supplied with...Ch. 2 - If E. coli were grown in a medium containing the...Ch. 2 - Prob. 4MCQCh. 2 - Prob. 5MCQCh. 2 - Prob. 6MCQCh. 2 - The dissociation products of the molecules are...Ch. 2 - Prob. 8MCQCh. 2 - The dissociation products of the molecules are...Ch. 2 - Prob. 10MCQCh. 2 - When you blow bubbles into a glass of water, the...Ch. 2 - Prob. 2ACh. 2 - Prob. 3ACh. 2 - Prob. 4ACh. 2 - Prob. 1CAECh. 2 - Prob. 2CAECh. 2 - Newborn babies are tested for phenylketonuria...Ch. 2 - The antibiotic amphotericin B causes leaks in...Ch. 2 - Prob. 5CAE
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biology and related others by exploring similar questions and additional content below.Similar questions
- Which of the following is NOT a unit of concentration? All of these options are units of concentration. mM Moles/Liter Molar You gathered your data from Lab , and found the following values: Total Activity of lysozyme in HEW: 14 units Total Activity of lysozyme in carb 1: 21 units Total amount of protein in HEW: 70 mg Total amount of protein in Carb 1: 15 mg Calculate the extent of purification of lysozyme in Carb 1. A 7 fold B 150% C 15 fold D 70%arrow_forwardName the protein structural element shown in yellow in the figure: B-barrel reverse turn disordered region a-helix parallel ß sheetarrow_forwardMatch the following levels of protein structure with their description two or more polypeptide chains > > twisting and folding to form a 3-dimensional structure, dependent on the locations of the charged groups and polar groups within the amino acid chain, sometimes covalent bonds form between sulfur atoms (called disulfide bonds) alpha helix and beta- pleated sheets formed by the arrangement of hydrogen bonds between amino acids, many other shapes are possible amino acid sequence 1. quaternary 2. tertiary 3. primary 4. secondaryarrow_forward
- Draw a hydropathy plot and a 2D protein cartoon for a protein that passes through the lipid bilayer 6 times..arrow_forwardDraw the chair conformation of the whole of O-linked oligosaccharide pictured below, including the amino acid it is attached to.arrow_forward1) What are the four levels of protein folding. How do you distinguish those different levels? What can denature a protein? 2) What are detergents and why are they useful? How do they basically work? 3) What is meant by amphipathic? What is an example of this?arrow_forward
- A tetradecapeptide (14 amino acid residues) gives the following peptide fragments on partial hydrolysis. From this information, deduce the primary structure of this polypep- tide. Fragments are grouped according to size. Pentapeptide Fragments Tetrapeptide Fragments Phe-Val-Asn-Gln-His Gln-His-Leu-Cys His-Leu-Cys-Gly-Ser His-Leu-Val-Glu Gly-Ser-His-Leu-Val Leu-Val-Glu-Alaarrow_forwardWhich of the following statement is not true? In a secondary structure the consecutive residues have phi and psi values that are quite different. The Ramachandran diagram defines the restrictions of polypeptide backbone conformation. Two α helices with hydrophobic side chains repeated in a regular pattern can form dimeric structures in water. Greek key motifs occur frequently in antiparallel β structures. Both α helices and β sheets can be amphipathic.arrow_forwardWhich of the following best describes how the secondary structure of a protein is formed? A B с D O=U a-helix H O=C N-H R-C-H C=0 H-N H-C-R O=C N-H R-C-H C=O H-N O-C N-H R-C-H C=O H-N N-H 0= H-C-R H-N C=O R-C-4 N-H O=C H-C-R 4-1 Ç=O R-C-H N-H 0=C H-C-R (=O R-C-H B-pleated sheet ionic bonds between the R groups of the polypeptide amino acids -Uh hydrogen bonds between the carboxyl and amino groups of non-adjacent amino acids covalent bonds between the carboxyl and amino groups of adjacent amino acids hydrogen bonds between the R groups of the polypeptide amino acidsarrow_forward
- Draw the structure of the tetrapeptide MIEL. Refer to the slide sets for the structures of the amino acids. Encircle all the peptide bonds. Box and label the N- and C-terminal amino acid residues.arrow_forwardGive the force of interaction involved in the protein folding of the protein structure A and C: COO CH,C-N-H• • •0- Pleated sheet structure A H C Helical structure CH3 CH3 - CH3 -CHCH,CH, CH- CH, CH3 (CH,),NH, -0-CCH,- C=0..•HN CH, CH, CH,CH CH,CH CH, CH, CH OH 0=C -CH,-S,S-CH,- H,Ñ Structure A: H-bonding: Structure C: Van der Waals O Structure A: Covalent interaction; Structure C: dipole-dipole interaction O Structure A: Salt bridge: Structure C: H-bonding Structure A: Hydrophobic interaction; Structure C: ionic interaction 00000 Barrow_forwardHow many amino acids are present on the helix to the right? Any partially drawn amino acids count as one whole amino acid Circle one peptide bond and box one R group on the figure to the right Describe how a lack of Vitamin C in the diet will affect the overall structure of collagen. Figure 6-7 Illustration, Irving Gels. Image from the Irving Geis Collection/Howard Hughes Medical Institute. Rights owned by HHMI. Reproduction by permission only. Drotoincarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

Biomolecules - Protein - Amino acids; Author: Tutorials Point (India) Ltd.;https://www.youtube.com/watch?v=ySNVPDHJ0ek;License: Standard YouTube License, CC-BY