
Study Guide for Campbell Biology
11th Edition
ISBN: 9780134443775
Author: Lisa A. Urry, Michael L. Cain, Steven A. Wasserman, Peter V. Minorsky, Jane B. Reece, Martha R. Taylor, Michael A. Pollock
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 2, Problem 10IQ
Look at your sketch of a water molecule in Interactive Question 2.9. Why is it roughly V-shaped?
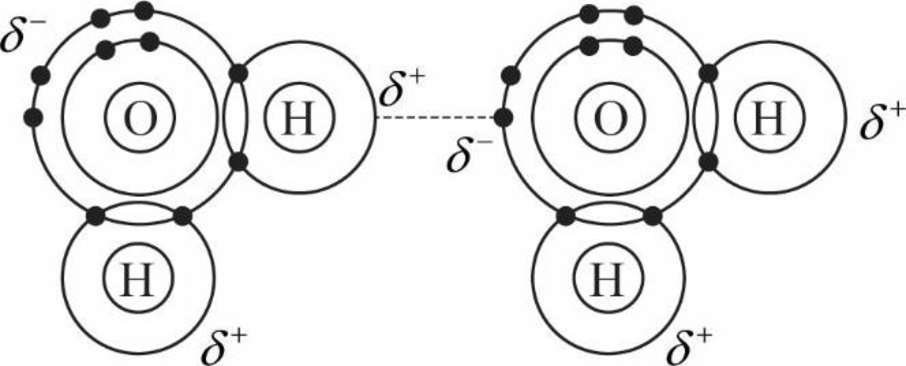
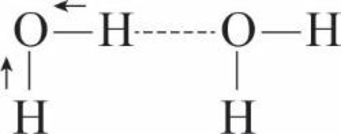
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Examine the water molecule in figure 2.14 of the textbook . What type of bond occurs between each hydrogen and the oxygen
Lipids are soluble in --- and insoluble in ---, respectively.
Question 3 options:
organic solvents, water
water, organic solvents
In your own words, list and define 3 properties of water that make it essential to life.
Chapter 2 Solutions
Study Guide for Campbell Biology
Ch. 2 - Fill in the names beside the symbols of the...Ch. 2 - The difference between the mass number and the...Ch. 2 - To move to a shell farther from the nucleus, an...Ch. 2 - Prob. 4IQCh. 2 - Fill in the blanks in the following concept map to...Ch. 2 - Prob. 6IQCh. 2 - Prob. 7IQCh. 2 - Prob. 8IQCh. 2 - Draw the structural formula of a water molecule,...Ch. 2 - Look at your sketch of a water molecule in...
Ch. 2 - Prob. 11IQCh. 2 - Prob. 1SYKCh. 2 - Atoms can have various numbers associated with...Ch. 2 - Prob. 3SYKCh. 2 - Prob. 1TYKCh. 2 - Prob. 2TYKCh. 2 - Prob. 3TYKCh. 2 - Prob. 4TYKCh. 2 - Radioactive isotopes can be used in studies of...Ch. 2 - Prob. 6TYKCh. 2 - Prob. 7TYKCh. 2 - Prob. 8TYKCh. 2 - Prob. 9TYKCh. 2 - Prob. 10TYKCh. 2 - Prob. 11TYKCh. 2 - Prob. 12TYKCh. 2 - Prob. 13TYKCh. 2 - Prob. 14TYKCh. 2 - Prob. 15TYKCh. 2 - A covalent bond between two atoms is likely to be...Ch. 2 - Prob. 17TYKCh. 2 - Prob. 18TYKCh. 2 - For questions 19-21, choose from the following...Ch. 2 - Prob. 20TYKCh. 2 - Prob. 21TYKCh. 2 - Prob. 22TYKCh. 2 - Prob. 23TYKCh. 2 - Prob. 24TYKCh. 2 - Prob. 25TYKCh. 2 - What is the difference between a molecule and a...Ch. 2 - Prob. 27TYKCh. 2 - Prob. 28TYK
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biology and related others by exploring similar questions and additional content below.Similar questions
- Consider molecule #1. How would these molecules arrange themselves when added to water? A. Would dissolve in water B. Would form a micelle C. Would form a bilayerarrow_forwardHow many moles of ATP can be obtained from palmitic acid? Show your solution.arrow_forwardThe questions in the left hand column correspond to the diagram in the right hand column. Read each question carefully and transfer your answers to the answer page provided. 1. Name this molecule. Be specific. CH,OH CH,OH 2. What two monomers make up the molecule? List in order from left to right. Be specific with the name. он Он OHarrow_forward
- Using the image attached: What is the electron geometry of this molecule?arrow_forwardThe highlighted structure is what? #3arrow_forwardWhat pH range is acidic? basic? What pH is water? What is a buffer? Read Chapter Summary for 2.3 and 2.4 Test Yourself on page 45-46 5. 6. Apply what you know on page 46 5. Section 2.5 Organic molecules – Why is carbon the common building block of all organic molecules? Figure 2.13 Dehydration synthesis and hydrolysis Breakdown both terms: "Dehydration" – removal of water "Synthesis" – to build "Нydro - " - water "- lysis" – to break apart Section 2.6 Carbohydrate – used for and Carbo" – means and -hydrate means Monosaccharide – pg. 5arrow_forward
- When reacted, how does the use of potassium (K+) vs sodium (Na+) change the soap product? Question 26 options: Na+ adds jasmine fragrance and K+ adds a rosemary fragrance to the soap. K+ produces harder, moldable soaps into shapes and Na+ produces softer, liquid soaps. There is no difference between the two cations. Na+ produces harder, moldable soaps into shapes and K+ produces softer, liquid soaps.arrow_forwardPhospholipid molecules were suspended in a mixture made up of water/oil. Describe the molecular arrangement that will occur and provide explanation for the arrangement of the molecules in the aqueous/oil mixture environment and its application in cell membrane functionsarrow_forwardHighlight each glycosidic bond in the molecule below. Then answer the questions in the table under the drawing area. HO-CH, H но Но HO–CH, H H ОН Н H ОН ن H H ОН Н ОН ОН H CH, H ОН H H ОН H но-сн, Н H ОН H он H ОН ОНarrow_forward
- ________ atoms give organic molecules their overall shape; ________ atoms determine the overall chemical behavior of organic molecules.arrow_forwardThree pigment solutions are given to you. The first pigment, A, contains four -OH and two -NH groups. The second pigment, B, has two-OH groups. The third pigment, C, has a very long hydrocarbon chain with no oxygen. You are told to separate these pigments by paper chromatography using a solvent composed of 70% benzene and 30% acetone. Predict the locations of the three pigments on the paper with respect to the solvent front after they separate. Explain your answer.arrow_forwardWhich image is an Organic molecule and which one is a inorganic molecule?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON
Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax
Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education,
Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education, Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company
Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co.
Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co. Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education
Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education

Human Anatomy & Physiology (11th Edition)
Biology
ISBN:9780134580999
Author:Elaine N. Marieb, Katja N. Hoehn
Publisher:PEARSON

Biology 2e
Biology
ISBN:9781947172517
Author:Matthew Douglas, Jung Choi, Mary Ann Clark
Publisher:OpenStax

Anatomy & Physiology
Biology
ISBN:9781259398629
Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa Stouter
Publisher:Mcgraw Hill Education,

Molecular Biology of the Cell (Sixth Edition)
Biology
ISBN:9780815344322
Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter Walter
Publisher:W. W. Norton & Company

Laboratory Manual For Human Anatomy & Physiology
Biology
ISBN:9781260159363
Author:Martin, Terry R., Prentice-craver, Cynthia
Publisher:McGraw-Hill Publishing Co.

Inquiry Into Life (16th Edition)
Biology
ISBN:9781260231700
Author:Sylvia S. Mader, Michael Windelspecht
Publisher:McGraw Hill Education
GCSE Chemistry - Acids and Bases #34; Author: Cognito;https://www.youtube.com/watch?v=vt8fB3MFzLk;License: Standard youtube license