
Concept explainers
(a) Deduce the structure of product A, which is highly symmetrical:
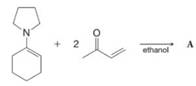
The following are selected spectral data for A:
MS
IR
(b) Write a mechanism that explains the formation of A.
Want to see the full answer?
Check out a sample textbook solution
Chapter 19 Solutions
Organic Chemistry
Additional Science Textbook Solutions
Anatomy & Physiology (6th Edition)
Genetic Analysis: An Integrated Approach (3rd Edition)
Campbell Essential Biology (7th Edition)
Applications and Investigations in Earth Science (9th Edition)
Chemistry: An Introduction to General, Organic, and Biological Chemistry (13th Edition)
Microbiology: An Introduction
- The complex anion in Ba₂[Cr(CN)6] is a tetragonally distorted octahedral complex (Dan). Baz[Cr(CN)6] is paramagnetic at room temperature with S = 1. Assume that the complex is a low-spin complex. a) Identify if the [Cr(CN)6] anionic complex has 4 long and 2 short bonds (left side of figure) or if the complex has 4 short and 2 long bonds (right side of figure) with respect to Oh symmetry. Use crystal field theory to answer this question. Explain/rationalize your decision. Can the provided information decide on the order of orbital energies? Dah Tetragonal Distortion ய Dab z-compression z-elongation x and y elongation O symmetry x and y compression E eg d² dx²-y² t2g dxy dxz dyz Question 4 a) continued: Provide your explanations in the space below. b) At low temperatures Ba₂[Cr(CN)6] is ferromagnetically ordered with a phase transition to a paramagnetic phase at Tc = 150K. Sketch the magnetic susceptibility vs. temperature in the diagram below. Indicate Tc as well as the paramagnetic and…arrow_forwarda) Draw the octahedral mer-[FeCl3(CN)3] complex and determine its point group. Use proper wedges and dashes in order to illustrate 3 dimensional details. Use the point group to determine if the complex has a resulting net dipole moment and describe its allowed direction with respect to its symmetry elements (if applicable). ード M 4- b) Substitute one chlorido ligand in mer-[FeCl3(CN)3] 4 with one fluorido ligand. Determine all possible isomers and their corresponding point groups. Use the point groups to determine if the complexes have resulting net dipole moments and describe their allowed direction with respect to its symmetry elements (if applicable). The number of complex sketches below is not necessarily indicative of the number of isomers. 4- 4- ☐☐☐ c) Substitute two chlorido ligands in mer-[FeCl3 (CN)3] 4 with two fluorido ligands. Determine all possible isomers and their corresponding point groups.. Use the point groups to determine if the complexes have resulting net dipole…arrow_forwardShow work. don't give Ai generated solutionarrow_forward
- Differentiate electron spin and electron spin moment.arrow_forwardDifferentiate between nuclear spin and electron spin.arrow_forwardDraw the trigonal prismatic MH6 molecular compound, where M is a 3d transition metal. a) Draw the trigonal prismatic MH6 molecular compound and determine its point group. b) i. What is the symmetry species for the 4s orbital on the central metal? ii. What is the symmetry species for the 3dx²-y² orbital on the central metal? Note: The z-axis is the principal axis. iii. Suggest a crystal field energy diagram for a d² electron configuration in a trigonal prismatic coordination environment. Label the metal d-orbital with their corresponding symmetry species label. Use the appropriate character table in the resource section.arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

