
Interpretation:
The two types of bonding which will occur in the bonding of the enzyme-substrate complex should be given with dotted lines.
Concept Introduction:
Enzyme:
- It is a protein or a molecule which can act as a catalyst for a biological reaction.
- Does not affect the equilibrium point of the reaction.
- Active site of the enzyme is the region where the reaction takes place.
- Enzyme’s activity can be specific which means the activity is limited to a certain substrate and a certain type of reaction and it is referred to as specificity of the enzyme.
Hydrogen bonding: It is an unusual strong intermolecular force occurs between a hydrogen atom and an electronegative atom like nitrogen, oxygen or fluorine. It occurs in both water and ammonia
Answer to Problem 19.24UKC
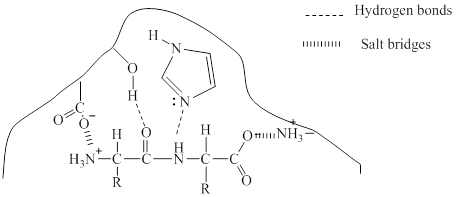
Explanation of Solution
Enzymes are three dimensional molecule with a catalytic site into which the substrate can fit. The enzymes are said to be so specific in their action because the activity is limited to a certain substrate and a certain type of reaction and it is referred to as specificity of the enzyme. It can be said that few molecules have the appropriate shape and functional groups to fit into the catalytic site.
Given in the diagram a dipeptidase enzyme and the substrate to form the enzyme-substrate complex. Different types of bonding may occur between the enzyme and the substrate to form the enzyme-substrate complex.
Hydrogen bonding is the attraction between a hydrogen atom and an electronegative atom and it can be considered as a special type of dipole-dipole interaction. The oxygen atom in the substrate can make hydrogen bond with hydrogen in the enzyme.
Salt bridges are formed between oppositely charged residues that are nearby to experience electrostatic attraction. Here, salt bridge is formed between the anionic carboxylate ion
The bonding is shown below:
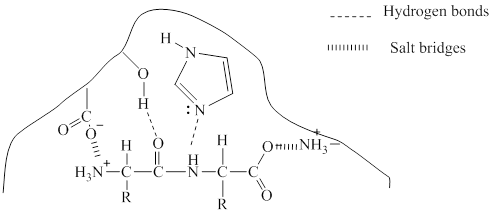
The two types of bonding which will occur in the bonding of the enzyme-substrate complex is given with dotted lines.
Want to see more full solutions like this?
Chapter 19 Solutions
Fundamentals of General, Organic, and Biological Chemistry (8th Edition)
- How many moles of active sites are there in 1 mg of enzyme? Assume that each subunit has one active site.arrow_forwardIn an enzyme's active site, a catalytic histidine residue abstracts a proton from the hydroxyl group of a catalytic serine residue. The catalytic serine then performs a nucleophilic attack on the carbonyl carbon of a peptide bond (on the substrate). The final products of this reaction are a cleaved substrate at the peptide bond, with a new amino group and carboxyl group, and regenerated catalytic residues. In this reaction, the serine residue is displaying what? A. Proximity and Orientation B. Covalent catalysis C. Metal ion catalysis D .A and B E. B and C F. None stated herearrow_forwardWhen studying the mechanism of the enzymatic reaction, functional groups were found that ensure the connection of the enzyme molecule with the substrate and take a direct part in the act of catalysis. What are these areas of the enzyme formed by these groups called? What functional structures form them and why?arrow_forward
- Can same enzyme have different kinetics for different substrates? What is the biological significance that?arrow_forwardHow is it possible to determine the structure of an enzyme substrate complex by x ray crystallography when the reaction is over so quickly and the x ray analysis takes at least several minutes?arrow_forwardWhat would be the result of an enzyme having a greater binding energy for the substrate than for the transition state?arrow_forward
- What happens to a denatured enzyme regarding its functionality? How can that result be explained with the help of the lock and key model?arrow_forwardwhich of the following describes an enzyme ALLOSTERIC site?arrow_forwardWhy might the compound shown below act as a transition state analog of phosphoglucose isomerase? A drawing of the normal transition state for this enzyme is needed.arrow_forward
- What metabolic substrate is produced from the carbon atoms of each of the following amino acids?Spell out the full name of the compound. 1) isoleucine 2) asparagine 3) threonine 4) argininearrow_forwardEach of the amino acid residues in the catalytic triad is part of what polypeptide chain?arrow_forwardWhich of the following statements helps best explain the reaction specificity of an enzyme? a) The shape and charge of the substrates are compatible with the active site of the enzyme. b) The free energy of the reactants is greater than the free energy of the products. c) The equilibrium constant of the reaction is much greater than 1. d) The concentration of the enzyme inside living cells is greater than the concentration of substrate.arrow_forward
 BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman
BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman
Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY
Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON
Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON





