
Organic Chemistry (8th Edition)
8th Edition
ISBN: 9780134042282
Author: Paula Yurkanis Bruice
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 18, Problem 78P
- a. Describe four ways the following reaction can be earned out:
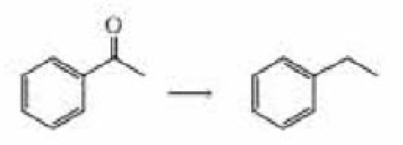
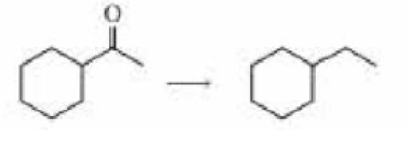
- b. Describe three ways the following reaction can be carried out:
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
These reagents can produce ketones with alkynes
A. BH3, THF, H2O2
B. KMnO4
C. O3
D. H2SO4, H2O, HgSO4
choices:A,DB,CA,B,CA,B,C,D
a.
b.
C.
d.
e.
type:
OH
type:
type:
OH
2
+
OH
TsOH cat.
HÅH
н Ἡ
Acetal formation
OH
TsOH cat.
SOCI₂
ܘ ܀
1.2 equiv. NH3
2. LIAIH₂
3. H₂O*
Which reaction can be used to carry out the following transformation?
A.
B. 1.
AICI 3
AIC13
2. HCI, Zn(Hg)
C. Either is okay
D. Neither will work
Chapter 18 Solutions
Organic Chemistry (8th Edition)
Ch. 18.1 - Draw the structure for each of the following: a....Ch. 18.3 - Why does hydration inactivate FeBr3?Ch. 18.6 - Prob. 4PCh. 18.7 - What is the major product of a Friedel-Crafts...Ch. 18.9 - Describe two ways to prepare each of the following...Ch. 18.10 - Prob. 7PCh. 18.11 - Name the following:Ch. 18.11 - Draw a structure for each of the following: a....Ch. 18.11 - Draw the structure for each of the following: a....Ch. 18.11 - Correct the following incorrect names: a....
Ch. 18.12 - Prob. 14PCh. 18.12 - List the compounds in each set from most reactive...Ch. 18.13 - Prob. 16PCh. 18.13 - What product(s) result from nitration of each of...Ch. 18.13 - Prob. 18PCh. 18.13 - What products are obtained from the reaction of...Ch. 18.15 - Give the products, if any, of each of the...Ch. 18.16 - a. Does a coupling reaction have to be used to...Ch. 18.16 - Show how the following compounds can be...Ch. 18.16 - Prob. 24PCh. 18.17 - What is the major product(s) of each of the...Ch. 18.17 - Prob. 26PCh. 18.18 - Why isn't FeBr3 used as a catalyst in the first...Ch. 18.18 - Prob. 29PCh. 18.18 - Write the sequence of steps required for the...Ch. 18.18 - Show how the following compounds can be...Ch. 18.19 - What product is formed from reaction of...Ch. 18.19 - Prob. 33PCh. 18.19 - Draw the structure of the activated ring and the...Ch. 18.20 - Prob. 35PCh. 18.20 - Prob. 36PCh. 18.20 - Diazomethane can be used to convert a carboxylic...Ch. 18.21 - Prob. 38PCh. 18.21 - Prob. 39PCh. 18.21 - Prob. 40PCh. 18.22 - Prob. 41PCh. 18 - Draw the structure for each of the following: a....Ch. 18 - Name the following:Ch. 18 - Prob. 44PCh. 18 - Prob. 45PCh. 18 - For each of the statements in Column I, choose a...Ch. 18 - What product is obtained from the reaction of...Ch. 18 - Draw the product(s) of each of the following...Ch. 18 - Rank the following substituted anilines from most...Ch. 18 - Prob. 50PCh. 18 - Prob. 51PCh. 18 - Show how the following compounds can be...Ch. 18 - Prob. 53PCh. 18 - The compound with the 1H NMR spectrum shown below...Ch. 18 - Rank each group of compounds from most reactive to...Ch. 18 - Prob. 56PCh. 18 - Prob. 57PCh. 18 - For each of the following components, indicate the...Ch. 18 - Prob. 59PCh. 18 - Prob. 60PCh. 18 - Describe two ways to prepare anisole from benzene.Ch. 18 - Prob. 62PCh. 18 - The following tertiary alkyl bromides undergo an...Ch. 18 - An aromatic hydrocarbon with a molecular formula...Ch. 18 - Show how the following compounds can be...Ch. 18 - Use the four compounds shown below to answer the...Ch. 18 - a. Rank the following esters from most reactive to...Ch. 18 - A mixture of 0.10 mol benzene and 0.10 mol...Ch. 18 - Prob. 69PCh. 18 - Prob. 70PCh. 18 - Benzene underwent a Friedel-Crafts acylation...Ch. 18 - Prob. 72PCh. 18 - Prob. 73PCh. 18 - Friedel-Crafts alkylations can be carried out with...Ch. 18 - Show how the following compounds can be prepared...Ch. 18 - Prob. 76PCh. 18 - Prob. 77PCh. 18 - a. Describe four ways the following reaction can...Ch. 18 - Propose a mechanism for each of the following...Ch. 18 - How can you prepare the following compounds with...Ch. 18 - Describe how naphthalene can he prepared from the...Ch. 18 - Using resonance contributors for the carbocation...Ch. 18 - Prob. 83PCh. 18 - What reagents are required to carry out the...Ch. 18 - Prob. 85PCh. 18 - Prob. 86PCh. 18 - Prob. 87PCh. 18 - Propose a mechanism for each of the following...Ch. 18 - P-Fluoronitrobenzene is more reactive toward...Ch. 18 - When heated with chromic acid, compound A forms...Ch. 18 - Show how the following compounds can be prepared...Ch. 18 - How can you distinguish the following compounds...Ch. 18 - Describe how mescaline can be synthesized from...Ch. 18 - Propose a mechanism for the following reaction...Ch. 18 - Propose a mechanism for each of the following...Ch. 18 - Describe how 3-methyl-1-phenyl-3-pentanol can he...Ch. 18 - An unknown compound reacts with ethyl chloride and...Ch. 18 - a. Explain why the following reaction leads to the...Ch. 18 - Explain why hydroxide ion catalyzes the reaction...Ch. 18 - Prob. 100PCh. 18 - Prob. 101PCh. 18 - a. How can aspirin be synthesized from benzene? b....Ch. 18 - Prob. 103PCh. 18 - Show how Novocain, a painkiller used frequently by...Ch. 18 - Prob. 105PCh. 18 - Saccharin, an artificial sweetener, is about 300...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Which product shown is the correct product formed in this multi-step reaction? Acetic Acid Chloride so, Benzene AICI, H,SO, to a bito SO,H so,H CO.H So,H D. B A B.arrow_forwardWhat is the final product of the following reaction? A. B. CI میں NaOH, Cl2 (excess C. D. ہیں CI مل CI OHarrow_forwardDetermine which reactions would occur (substitution, elimination, both, or neither). CH;CH,OH a. SN1 O b. E1 C. SN1 & E1 O d. SN2 е. Е2 O f. SN2 & E2 g. No reactionarrow_forward
- 48. Which set of reagents would accomplish this conversion H. H. CH3OH with H* as a catalyst NaH followed by CH3OH CH3ONA followed CH3OH CH3ONA followed by CH3Br a. b. C. d.arrow_forward1. Draw the products of each nucleophilic substitution reaction a. b. D b OH C. d. e. f. Br 1 NaCN + NaOCH3 H₂Oarrow_forwardSynthesis 10.63 Devise a synthesis of each product from the given starting material. More than one step is required. a. b. d. e. Br Br OH OCH3 ta CI OHarrow_forward
- 2. Draw the products of each reaction. CH;CH2CI b. HNO3 Cl2 OCH3 Br -NO2 a. с. AICI3 H,SO4 FeCl3 3. Draw the products formed when each compound is treated with HNO3 and H2SO4. State whether the reaction occurs faster or slower than a similar reaction with be nzene.arrow_forwardPredict the product of each reaction or sequence of reactions. a) b) Na₂Cr₂O7 H₂SO4, H₂O 1. NBS, A 2. t-BUOK 1. NBS, A 2. NACECH e) 1. NBS, A 2. t-BuOK 3. HCI 1. NBS, A 2. Mg O 3.arrow_forward1. Provide the major product for the following reactions. If the reaction creates a racemic mixture, draw only one of the stereoisomers (meaning put wedge and dash bonds on the structure). a. b. C. ZI OMe OH LOTS NaCN DMFarrow_forward
- Oraw a stepwise mechanism for the following reaction: Br CH3CH2OH Part 1 out of 3 HBr Br Br CH3CH,Br finish structure ... draw structure CH;Brarrow_forward5. What reagents are needed to convert toluene (C,H,CH,) to each compound? a. C.H.COOH b. C.H₂CH₂Br c. p-bromotoluene d. o-nitrotoluene e. p-ethyltoluene f.arrow_forward10. What products are formed when the following compound is treated with each reagent? If no reaction occurs, write NR. a. H₂/Pd b. K₂Cr2O7 c. Tollen's reagent d. 2 equiv. CH3OH, H+ e. 2 equiv. CH3CH₂OH, H+ f. Product of (e), then H₂O/H*arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
General Chemistry | Acids & Bases; Author: Ninja Nerd;https://www.youtube.com/watch?v=AOr_5tbgfQ0;License: Standard YouTube License, CC-BY