
Organic Chemistry (8th Edition)
8th Edition
ISBN: 9780134042282
Author: Paula Yurkanis Bruice
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 18, Problem 46P
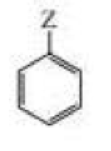
For each of the statements in Column I, choose a substituent from Column II that fits the description for the compound on the right:
| Column I | Column II |
| a. Z donates electrons by hyperconjugation and does not donate or withdraw electrons by resonance. | OH |
| b. Z withdraws electrons inductively and withdraws electrons by resonance. | Br |
| c. Z deactivates the ring and directs ortho-para. | +NH3 |
| d. Z withdraws electrons inductively, donates electrons by resonance, and activates the ring. | CH2CH3 |
| e. Z withdraws electrons inductively, and does not donate or electrons by resonance. | NO2 |
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
10. A compound X of molecular formula C8H12 with no triple bonds reacts with one
equivalent of H2 to give a new compound having molecular formula CaH14.
What can be inferred about the structure of compound X?
A) Compound X has 3 rings.
B)
Compound X has 3 pi bonds.
Compound X has 1 ring and 2 pi bonds.
D) Compound X has 2 rings and 1 pi bond.
11. Determine the product of the following reaction.
H,
Lindlar's cat.
%3D
IV
A)I B) II C) III D) IV
Type here to search
互i | A C
三D
C Chemistry
nardi and Neil Schore
presented by Sap
Propose an efficient synthetic scheme for the conversion of lactone A into amine B, a precursor to the
naturally occurring monoterpene C. Note: If one or more reagents are incorrectly placed, a single red X will
appear on the top left.
C
CHs
CHs
CH3
2.
CH3
H3C
CH3
H3C
CHs
CH3
H3C
CH
но
но
3.
CH3
Question 2 of 8
(c)
ok
CH3COgH
Product(s):
* Draw Your Solution
Save for Later
MacBoo
esc
80
F3
FI
F2
DOO
F4
Chapter 18 Solutions
Organic Chemistry (8th Edition)
Ch. 18.1 - Draw the structure for each of the following: a....Ch. 18.3 - Why does hydration inactivate FeBr3?Ch. 18.6 - Prob. 4PCh. 18.7 - What is the major product of a Friedel-Crafts...Ch. 18.9 - Describe two ways to prepare each of the following...Ch. 18.10 - Prob. 7PCh. 18.11 - Name the following:Ch. 18.11 - Draw a structure for each of the following: a....Ch. 18.11 - Draw the structure for each of the following: a....Ch. 18.11 - Correct the following incorrect names: a....
Ch. 18.12 - Prob. 14PCh. 18.12 - List the compounds in each set from most reactive...Ch. 18.13 - Prob. 16PCh. 18.13 - What product(s) result from nitration of each of...Ch. 18.13 - Prob. 18PCh. 18.13 - What products are obtained from the reaction of...Ch. 18.15 - Give the products, if any, of each of the...Ch. 18.16 - a. Does a coupling reaction have to be used to...Ch. 18.16 - Show how the following compounds can be...Ch. 18.16 - Prob. 24PCh. 18.17 - What is the major product(s) of each of the...Ch. 18.17 - Prob. 26PCh. 18.18 - Why isn't FeBr3 used as a catalyst in the first...Ch. 18.18 - Prob. 29PCh. 18.18 - Write the sequence of steps required for the...Ch. 18.18 - Show how the following compounds can be...Ch. 18.19 - What product is formed from reaction of...Ch. 18.19 - Prob. 33PCh. 18.19 - Draw the structure of the activated ring and the...Ch. 18.20 - Prob. 35PCh. 18.20 - Prob. 36PCh. 18.20 - Diazomethane can be used to convert a carboxylic...Ch. 18.21 - Prob. 38PCh. 18.21 - Prob. 39PCh. 18.21 - Prob. 40PCh. 18.22 - Prob. 41PCh. 18 - Draw the structure for each of the following: a....Ch. 18 - Name the following:Ch. 18 - Prob. 44PCh. 18 - Prob. 45PCh. 18 - For each of the statements in Column I, choose a...Ch. 18 - What product is obtained from the reaction of...Ch. 18 - Draw the product(s) of each of the following...Ch. 18 - Rank the following substituted anilines from most...Ch. 18 - Prob. 50PCh. 18 - Prob. 51PCh. 18 - Show how the following compounds can be...Ch. 18 - Prob. 53PCh. 18 - The compound with the 1H NMR spectrum shown below...Ch. 18 - Rank each group of compounds from most reactive to...Ch. 18 - Prob. 56PCh. 18 - Prob. 57PCh. 18 - For each of the following components, indicate the...Ch. 18 - Prob. 59PCh. 18 - Prob. 60PCh. 18 - Describe two ways to prepare anisole from benzene.Ch. 18 - Prob. 62PCh. 18 - The following tertiary alkyl bromides undergo an...Ch. 18 - An aromatic hydrocarbon with a molecular formula...Ch. 18 - Show how the following compounds can be...Ch. 18 - Use the four compounds shown below to answer the...Ch. 18 - a. Rank the following esters from most reactive to...Ch. 18 - A mixture of 0.10 mol benzene and 0.10 mol...Ch. 18 - Prob. 69PCh. 18 - Prob. 70PCh. 18 - Benzene underwent a Friedel-Crafts acylation...Ch. 18 - Prob. 72PCh. 18 - Prob. 73PCh. 18 - Friedel-Crafts alkylations can be carried out with...Ch. 18 - Show how the following compounds can be prepared...Ch. 18 - Prob. 76PCh. 18 - Prob. 77PCh. 18 - a. Describe four ways the following reaction can...Ch. 18 - Propose a mechanism for each of the following...Ch. 18 - How can you prepare the following compounds with...Ch. 18 - Describe how naphthalene can he prepared from the...Ch. 18 - Using resonance contributors for the carbocation...Ch. 18 - Prob. 83PCh. 18 - What reagents are required to carry out the...Ch. 18 - Prob. 85PCh. 18 - Prob. 86PCh. 18 - Prob. 87PCh. 18 - Propose a mechanism for each of the following...Ch. 18 - P-Fluoronitrobenzene is more reactive toward...Ch. 18 - When heated with chromic acid, compound A forms...Ch. 18 - Show how the following compounds can be prepared...Ch. 18 - How can you distinguish the following compounds...Ch. 18 - Describe how mescaline can be synthesized from...Ch. 18 - Propose a mechanism for the following reaction...Ch. 18 - Propose a mechanism for each of the following...Ch. 18 - Describe how 3-methyl-1-phenyl-3-pentanol can he...Ch. 18 - An unknown compound reacts with ethyl chloride and...Ch. 18 - a. Explain why the following reaction leads to the...Ch. 18 - Explain why hydroxide ion catalyzes the reaction...Ch. 18 - Prob. 100PCh. 18 - Prob. 101PCh. 18 - a. How can aspirin be synthesized from benzene? b....Ch. 18 - Prob. 103PCh. 18 - Show how Novocain, a painkiller used frequently by...Ch. 18 - Prob. 105PCh. 18 - Saccharin, an artificial sweetener, is about 300...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Chapter 2 [References] H3C HB CH3 На, CH3 CH3 D H3C B НО Cholestanol differs from cholesterol only in the absence of a double bond in ring B. Draw the three-dimensional structure of cholestanol, and then determine the orientation of the following groups: #1: H at the junction of rings A & B with respect to ring A #2: Methyl at the junction of rings A & B with respect to ring BỊ #3: HВ O Iarrow_forward1. Oxazolines (five membered ring with N and O shown below) are an important functional group in organic chemistry because they can be used to help facilitate catalytic, enantioselective reactions during the synthesis of important pharmaceutical molecules. One way to make oxazolines is through the ring expansion of N-acylaziridines. Me Ph Nal Acetone Ph An N-Acylaziridine An oxazoline a. This reaction utilizes nucleophilic substitution reactions to get from the aziridine to the oxazoline. Provide a complete arrow pushing mechanism for this transformation. You do not have to draw it while talking. You can have the mechanism already drawn out and simply talk through the steps. If you choose to use ChemDraw, be sure it is very clear where your arrows are pointing. You may also draw it by hand (paper or tablet) and screenshare an image of your work while explaining what happens. You must talk through each step of your mechanism and use proper terms such as nucleophile and electrophile. b.…arrow_forwardCH3 Buli Br Compound C C13H160 CH3 Propose a structure for compound C and the product C13H160. • You do not have to consider stereochemistry. You do not have to explicitly draw H atoms. • Do not include lone pairs in your answer. They will not be considered in the grading. • Draw organic products only. Do not include counter-ions, e.g., Na, I, in your answer. • Draw one structure per sketcher. Add additional sketchers using the drop-down menu in the bottom right corner. Separate structures using the → sign from the drop-down menu.arrow_forward
- Consider the following compounds: 1. 1-hydroxy-3-heptanone III. 2-hydroxy-3-heptanone II. 1-methoxy-2-hexanone IV. 1-hydroxy-4-methyl-3-hexanone Which are position isomers? O Il and III I and II I and III none I and IVarrow_forwardWhat is the correct structure for p-bromotoluene? ملی موسی مم Select one: OA. I OB. IV OC. III OD. II CH, LOCH, OCH, IV CH₂arrow_forward6. Draw Hhe stricture of Compound Y. Compound Y 03 2. Zn, Hz0" t.arrow_forward
- From each pair, select the stronger base. Then draw the conjugate acid of the strong base below. CH3CH20 or CH3CEC • You do not have to consider stereochemistry. You do not have to explicitly draw H atoms. • You do not have to include lone pairs in your answer. C opy astearrow_forwardA certain hydrocarbon had the molecular formula C16H26 and contained two triple bonds. Ozonolysis gave CH3(CH2), CO₂H and HO₂CCH2CH2CH2CH2CO₂H as the only products. Draw a reasonable structure for this hydrocarbon. Click and drag to start drawing a structure. D:arrow_forwardCH=CHNO₂ (2-nitrovinyl)benzene Electrophilic substitution on (2-nitrovinyl)benzene occurs at the meta position. Draw resonance structures to show how the ring is electron-poor at the ortho and para positions. You do not have to consider stereochemistry. • Draw one structure per sketcher. Add additional sketchers using the drop-down menu in the bottom right corner. • Separate resonance structures using the symbol from the drop-down menu. 0 78 00-F karrow_forward
- -k ces Give the IUPAC name for each compound. CH3CH₂CHCH₂CH3 CH3 a. 3 methyl CH3 CH₂CH₂CH3 b. CH3CH₂CH₂-C-C-CH₂CH₂CH₂CH3 I H H (select) pentane CH₂CH3 c. H-C-CH₂-CHCH3 CH3 CH3 ㄱㄷ (select) ✓(select) (select) (select) (select) Varrow_forward1. The line structure of a natural product that was isolated from various shrubs is shown below. This natural substance is very unusual in that it is rather unstable and decomposes when heated above 130 °C. Suggest an explanation for this property. 2. The structure of a vitamin is shown below. H HH H H C C-H `N' H. H H H H H Identify two potentially nucleophilic atoms and two potentially electrophilic atoms in your structure, giving a very brief explanation in each case.arrow_forward3. Draw each reaction entirely using line structures and answer the questions below. Which one of the following two reactions is effective in the synthesis of 4-methyl-2-hexyne? Why is the other not effective? caroon y carling Br 1. CH₂CH₂CHCH, + CH₂C=CNa CH₂ 2. CH₂CH₂CHC=CNa + CH₂1 the equarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
General Chemistry | Acids & Bases; Author: Ninja Nerd;https://www.youtube.com/watch?v=AOr_5tbgfQ0;License: Standard YouTube License, CC-BY