
Concept explainers
(a)
Interpretation:
The structural formula for the principal product formed when benzamide is treated with the given reagent has to be drawn.
Concept introduction:
The normal reaction of an amide with water does not give good yield of product. As amides are least reactive
The reaction of water and amides in presence of acid gives a carboxylic acid as a product. The reaction equation is written as,
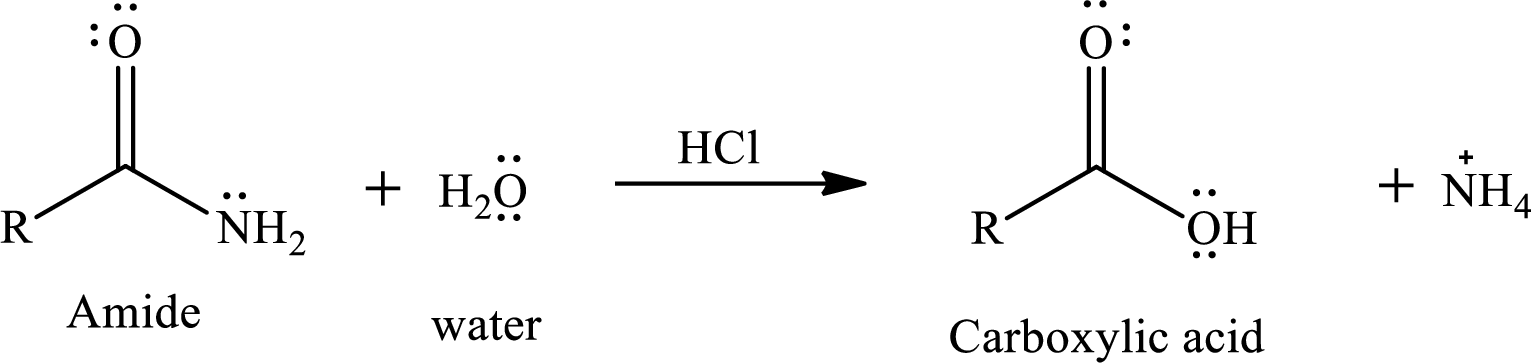
The reactivity of amide also depends upon the leaving tendency of amine part from acyl group. The weaker the base better will be the leaving group. The basicity of
(b)
Interpretation:
The structural formula for the principal product formed when benzamide is treated with the given reagent has to be drawn.
Concept introduction:
Amide hydrolysis in aqueous base:
A carboxylate salt and ammonia or an amine will be the product of amide hydrolysis in aqueous base.
For each mole of amide, one mole of base is required.
(c)
Interpretation:
The structural formula for the principal product formed when benzamide is treated with the given reagent has to be drawn.
Concept introduction:
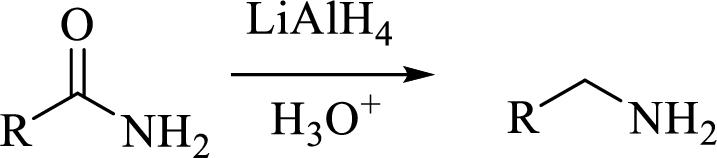
Trending nowThis is a popular solution!

Chapter 18 Solutions
Organic Chemistry, Loose-leaf Version
- Draw compounds that contain the following: (a) A primary alcohol (c) A secondary thiol (c) An isopropyl group (b) A tertiary nitrile (d) A quaternary carbonarrow_forward(a) Compound Z is a tertiary aromatic amine with the formula, C8H11N. Provide a chemical structure for compound Z. (b)nDraw the structure of the product formed exclusively when nitrous acid reacts with Z.arrow_forwardDescribe concisely a chemical test to distinguish between the following pairs ofcompounds. (a) Pentan-2-one and pentan-3-one(b) Propanal and propanone(c) Phenol and anilinearrow_forward
- 1. Draw structures corresponding to the following IUPAC names: (a) 4-Methylpentanoic acid (b) o-Hydroxybenzoic acid (c) 2,2-Dimethylpropanoyl chloride (d) trans-2-Methylcyclohexanecarboxamide (e) p-Methylbenzoic anhydride (f) p-Bromobenzonitrilearrow_forwardThe oxidation of 3-methylbutanal with potassium permanganate in an acid medium produces compound A and the reduction of cyclohexanone with NaBH4 produces compound B. Indicate the CORRECT alternative: a) A reacts rapidly with water to produce a carboxylic acid. b) Reaction of B with methylamine produces an amide. c) Reaction of A with an acyl chloride produces an ester. d) B reacts with HCN to produce a cyanohydrin. e) The reaction of A and B in an acid medium produces cyclohexyl 3-methylbutanoate.arrow_forwardDraw the structure of a compound of molecular formula C 9H 11NO that contains a benzene ring and a: (a) 1 ° amide; (b) 2 ° amide; (c) 3 ° amide.arrow_forward
- (a) Draw the structures for the eight constitutional isomers of molecular formula C 4H 11N. (b) Give the systematic name for each amine. (c) Identify the chirality center present in one of the amines.arrow_forwardDraw and name compounds that meet these descriptions:(a) Three different amides with the formula C5H11NO(b) Three different esters with the formula C6H12O2arrow_forwardPrepare CH3COOC2H5, ethyl acetate, using ethyl alcohol. Write the chemical reaction. The pain reliever acetaminophen is produced by reacting 4-aminophenol with acetic anhydride. Outline a synthesis of acetaminophen from 4-aminophenol including any needed inorganic reagents.arrow_forward
- Compounds that contain an N-H group associate by hydrogen bonding. (a) Do you expect this association to be stronger or weaker than that of compounds containing an O-H group? (b) Based on your answer to part (a), which would you predict to have the higher boiling point, 1-butanol or 1-butanamine?arrow_forwardQuestion -S1arrow_forwardEthyl butyrate, CH3CH2CH2CO2CH2CH3CH3CH2CH2CO2CH2CH3, is an artificial fruit flavor commonly used in the food industry for such flavors as orange and pineapple. Its fragrance and taste are often associated with fresh orange juice, and thus it is most commonly used as orange flavoring. It can be produced by the reaction of butanoic acid with ethanol in the presence of an acid catalyst (H+H+): CH3CH2CH2CO2H(l)+CH2CH3OH(l)H+⟶CH3CH2CH2CO2CH2CH3(l)+H2O(l) Part A Given 7.30 gg of butanoic acid and excess ethanol, how many grams of ethyl butyrate would be synthesized, assuming a complete 100%% yield? Express your answer in grams to three significant figures. Part B A chemist ran the reaction and obtained 5.95 gg of ethyl butyrate. What was the percent yield? Express your answer as a percent to three significant figures. Part C The chemist discovers a more efficient catalyst that can produce ethyl butyrate with a 78.0%% yield. How many grams would be produced from 7.30 gg of…arrow_forward
 Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning

