
Concept explainers
Give stereochemical formulas for compounds A–Q:
(a)
(b)
(c)
(d)
(e)
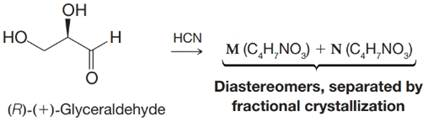
(f)
(g)
Want to see the full answer?
Check out a sample textbook solution
Chapter 17 Solutions
Organic Chemistry
Additional Science Textbook Solutions
CHEMISTRY-TEXT
Elementary Principles of Chemical Processes, Binder Ready Version
Organic Chemistry (8th Edition)
Introductory Chemistry (5th Edition) (Standalone Book)
Chemistry: The Molecular Nature of Matter
- Identify the functional groups in the following molecules. (Use names from the table below. List each class of functional group only once. If there are fewer than 3 functional groups, leave an appropriate number of answer boxes empty.) a) b) N- -CH3 Name Structure Alkene Arene Structures of Some Common Functional Groups Alkyne -C=C1 Halide ** Nitro Thiol Name Aldehyde Ketone Structure DO SH 0-0² 'H 0=0arrow_forwardIndicate the stereochemistry of the product of SN2 reaction: NaOH Br CH3 CH;OH Name the product:arrow_forwardRank the following groups in order of decreasing priority. −COOH, −CH2OH, −H, −CHOarrow_forward
- Identify the following reactions as either SN1, SN2, E1, or E2: (a) Br CHCH3 CH=CH2 кон (b) Br OCH3 .CHCH3 .CHCH3 CH3OH Heatarrow_forwardUnder certain reaction conditions, 2,3-dibromobutane reacts with two equivalents of base to give three products, each of which contains two new p bonds. Product A has two sp hybridized carbon atoms, product B has one sp hybridized carbon atom, and product C has none. What are the structures of A, B, and C?arrow_forwardPropose a synthesis for the systemic agricultural fungicide tridemorph from dodecanoic acid (lauric acid), propene, and a one-carbon building block. How many stereoisomers are possible for tridemorph? CH3 →CH3(CH2)10COOH + CH3CH=CH, H3C (CH2) 12CH3 Tridemorph Dodecanoic acid Propene (Lauric acid)arrow_forward
- Aldehydes and ketones react with thiols to yield thioacetals just as they react with alcohols to yield acetals. Predict the product of the following reaction, and propose a mechanism:arrow_forwardDraw structures corresponding to the following IUPAC names: (a) 1, 1-Dimethylcyclooctane (b) 3-Cyclobutylhexane (c) 1, 2-Dichlorocyclopentane (d) l, 3-Dibromo-5-methylcyclohexanearrow_forwardA chemist synthesized compound X as a racemic mixture. When the ketone group in X was enzymatically reduced to the corresponding alcohol, a 100% yield was obtained of the product shown below. Choose the statement that best describes this result. ОН enzyme C;H1 `OCH,CH; pH 4.0 C3H1 `OCH,CH3 ОН ÕH X (racemic) (100% yield) One enantiomer of compound X reacts quickly with the enzyme. The other enantiomer of compound X is unreactive, but rapidly equilibrates with the reactive enantiomer under the reaction conditions. Since compound X was racemic, it makes sense that only a single product was obtained. O The product is a meso compound, so either enantiomer of compound X gives the same product. One enantiomer of compound X reacts quickly with the enzyme, while the other enantiomer of compound X remains unchanged.arrow_forward
- Reaction of HBr with 2-methylpropene yields 2-bromo-2-methylpropane. What is the structure of the carbocation formed during the reaction? Show the mechanism of the reaction.arrow_forwardFor the substituted cyclohexane compound shown, identify the atoms that are trans to the bromo substituent. I CH3 Br J HD HCI НА FF HB НОЕ HG CI Identify the atoms that are trans to the bromine. A В D E F H Karrow_forwardMethyl isocyanate, CH3 -N= C = O, is used in the industrial synthesis of a type of pesticide and herbicide known as a carbamate. As a historical note, an industrial accident in Bhopal, India, in 1984 resulted in leakage of an unknown quantity of this chemical into the air. An estimated 200,000 people were exposed to its vapors, and over 2000 of these people died. Q.) Methyl isocyanate reacts with strong acids, such as sulfuric acid, to form a cation. Will this molecule undergo protonation more readily on its oxygen or nitrogen atom? In considering contributing structures to each hybrid, do not consider structures in which more than one atom has an incomplete octetarrow_forward
