
Essential Organic Chemistry, Global Edition
3rd Edition
ISBN: 9781292089034
Author: Paula Yurkanis Bruice
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 14.4, Problem 3P
Interpretation Introduction
Interpretation:
Number of hydrogen atoms which are attached to the secondary carbons in the given structure has to be determined.
Concept introduction:
In accordance with the bonds and the connection the carbon atom is an organic molecule can be classified into four and they are mentioned below,
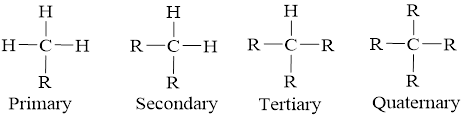
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Which carbon atom of this molecule have disstereotopic hydrogen?
How many secondary hydrogens are there in the following compound?
Hello, I hope you are doing well on this fine day. For the following quetion please read carefully the question and instruction. PLEASE ANSWER QUESTION IN 20 MINTUES NOT MORE PLEASE AND THANK YOU. If you do answer the question correctly and post it in the next 20 minutes, NO NEED TO SHOW ALL THE WORK, I JUST WOULD LIKE THE CORRECT ANSWER AS SOON AS POSSIBLE. I will write a wonderful and generous feedback/review/rating about you.
How many total sigma bonds are in the following molecule:
CH2CHCH3
Chapter 14 Solutions
Essential Organic Chemistry, Global Edition
Ch. 14.2 - Write the mechanism for the monobromination of...Ch. 14.2 - Prob. 2PCh. 14.4 - Prob. 3PCh. 14.4 - Which of the hydrogens in the structure in the...Ch. 14.4 - How many alkyl chlorides can be obtained from...Ch. 14.4 - Prob. 7PCh. 14.5 - Prob. 9PCh. 14.6 - a. Which ether is most apt to form a peroxide? b....Ch. 14.7 - Prob. 11PCh. 14 - Prob. 12P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- For the part circled in white, I don't understand when simplifying this why we don't divide the coefficient in front of octane by 2. I thought it would be 1/2 Since we divided the other ones by 2.arrow_forwardConsider the molecule whose line structure is below: 1. The molecule under discussion: a. has no stabilization due to delocalized π electrons b. is stabilized by delocalized π electrons 2. Identify the numbers of atoms that contribute to the delocalized orbitals. Use a comma as a separator. Arrange values from minimum to maximum. For consecutive values use "-" (for example: 1,3,5-10). If there are no delocalized orbitals enter "none".arrow_forwardExplanation for part C and D.arrow_forward
- This is not graded. Please, answer no. 22 & no. 23. Thank you.arrow_forwardplease help with the bond line structures and IUPAC name The ones done in blue are what I've answered - please check those for correctness The others I need help with...arrow_forward8. Please show the answer needed to draw in the space below. I need the final structurearrow_forward
- A. Answer as directed. 5 pts each. 1. Draw a molecule that contains sp, sp², and sp³ hybridized carbon atom. 2. Explain why 1,2-dichloroethyne is nonpolar while cis-1,2-dichloroethene is not. Provide illustrations to support your answer. B. Given the molecular formulae, provide the table with relevant details. Methylamine is already done for you.. Condensed Formula CH3NH2 (methylamine/ methanamine) Drawing and Hybridization Molecular Geometry Approximate Bond Angle(s) with Drawing Molecular Polarity H-N-H: 109.5° CH3 sp³ (around N) H CF4 '\' (around C) The angle formu (around Carbon-3) 10SY Polar " CH3 H Condensed Formula Drawing and Molecular Geometry Hybridization Approximate Bond Angle(s) with Drawing (around the rightmost Carbon) 4 CH,CH₂SH (around S) 5 CHOOCH (dimethyl peroxide) (around left side O) 6 CH₁C=N (around middle C) 7 CH,COOH 8 CH3COOH (around Carbon-1) (around Carbon-2) Molecular Polarityarrow_forwardWhat carbons are electron deficient and the electron rich? Justify with all the correct resonance structures. I have figured out 4 resonance structures and know that there are 6 i need to prove.arrow_forwardMolecular formula from a line structurearrow_forward
- Compare and contrast the Lewis structure of nitromethane and methylnitrare .write down at least two resonance firms .what can you say about the polarity and order of NO bonds in each substancearrow_forwardThe IR spectrum below is most likely a: Aldehyde, Alkane, Alkene, Alkyl Chloride, Alkynearrow_forwardHow would you develop a molecular orbital for a structure like buta-2-diene that has an anion on the first carbon and a cation on the fourth carbon? Also, how would MO look like for 2-dimethyl-1,3-pentadiene?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY
Mass Spectrometry; Author: Professor Dave Explains;https://www.youtube.com/watch?v=hSirWciIvSg;License: Standard YouTube License, CC-BY