
Essential Organic Chemistry, Global Edition
3rd Edition
ISBN: 9781292089034
Author: Paula Yurkanis Bruice
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 14.6, Problem 10P
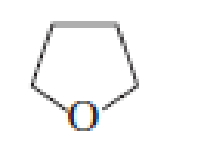
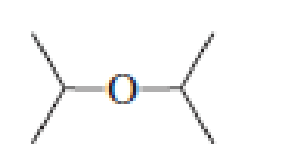
- a. Which ether is most apt to form a peroxide?
- b. Which ether is least apt to form a peroxide?
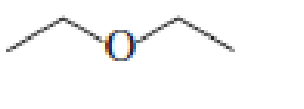
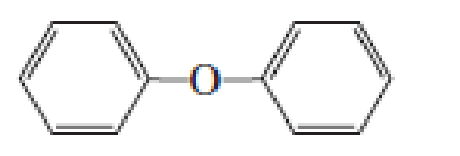
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
a. Which ether is most apt to form a peroxide?
b. Which ether is least apt to form a peroxide?
Synthesize each compound from acetylene. You may use any other organic or inorganic reagents.
C. CH,CH,CH,CH, CHO
d. CH3CH, CHỖ CH
Which ether is most apt to form a peroxide? Which ether is least apt to form a peroxide?
Chapter 14 Solutions
Essential Organic Chemistry, Global Edition
Ch. 14.2 - Write the mechanism for the monobromination of...Ch. 14.2 - Prob. 2PCh. 14.4 - Prob. 3PCh. 14.4 - Which of the hydrogens in the structure in the...Ch. 14.4 - How many alkyl chlorides can be obtained from...Ch. 14.4 - Prob. 7PCh. 14.5 - Prob. 9PCh. 14.6 - a. Which ether is most apt to form a peroxide? b....Ch. 14.7 - Prob. 11PCh. 14 - Prob. 12P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Provide all possible products formed in the given reaction Draw the products formed when each ether is treated with two equivalents of HBr.arrow_forward48. Which set of reagents would accomplish this conversion H. H. CH3OH with H* as a catalyst NaH followed by CH3OH CH3ONA followed CH3OH CH3ONA followed by CH3Br a. b. C. d.arrow_forward1. Draw the products of each nucleophilic substitution reaction a. b. D b OH C. d. e. f. Br 1 NaCN + NaOCH3 H₂Oarrow_forward
- O#1 Draw all possible constitutional isomers formed by DEHYDROHALOGENATION of each alkyl halide. Br CI Br b. а. С. d.arrow_forwardDraw the products formed when each ether is treated with two equivalents of HBr. a. (CH3),COCH,CH,CH3 b. С. FOCH3arrow_forwardWhich of the following molecules contain a? good leaving group Which of the following molecules contain a good leaving group? он a. b. C. d. OHarrow_forward
- Which statement is incorrect? A Grignard reagent is a strong lewis base a. O b. Water and alcohol decompose Grignard reagents Oc. Grignard reagents add to the carbonylic carbon of a ketone or aldehyde Ether and THF are solvents not suited for Grignard reactions. d.arrow_forward2. Draw the structures of the starting organic compounds that can be used to make the following ethers. ontarrow_forward16.69 Use the reactions in this chapter along with those learned in Chapters 11 and 12 to synthesize each compaund. You may use benzene, acetylene (HC=CH), ethanol, ethylene oxide, and any inorganic reagents. a. b. CI он C ON d. NO2arrow_forward
- Rank the compounds in each group in order of increasing water solubility. a. (CH3),CH, CH3OCH2CH3, CH3CH;CH,CH3, CH;CH,CH2OH b. Brarrow_forwardWhy are epoxides more reactive than other ethers (open chain and larger cyclic ethers)? Select one: A. It is easier for epoxides to undergo loss of water compared to other ethers. B. Epoxides are stronger bases than other ethers. C. Their reactions, a ring-opening process, relieves the strain of a three-membered ring D. Epoxides are more easily oxidized than other ethers.arrow_forward1 When epoxides react, they undergo ring-opening by a cleavage of the C-O bond. Why is it easier to break a C-O bond of an epoxide compared to breaking a C-O bond of other ethers? A. A more stable carbocation is formed when an epoxide ring-opens. B. Breaking the epoxide C-O bond relieves the strain of a three-member ring. C. Epoxide ring opening conforms to a faster E2 mechanism. D. Mechanistically, the epoxide carbon always becomes protonated, enhancing it's ring opening reactivity.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY
Alcohols, Ethers, and Epoxides: Crash Course Organic Chemistry #24; Author: Crash Course;https://www.youtube.com/watch?v=j04zMFwDeDU;License: Standard YouTube License, CC-BY