
Organic Chemistry - Standalone book
10th Edition
ISBN: 9780073511214
Author: Francis A Carey Dr., Robert M. Giuliano
Publisher: McGraw-Hill Education
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 14, Problem 50P
Figure
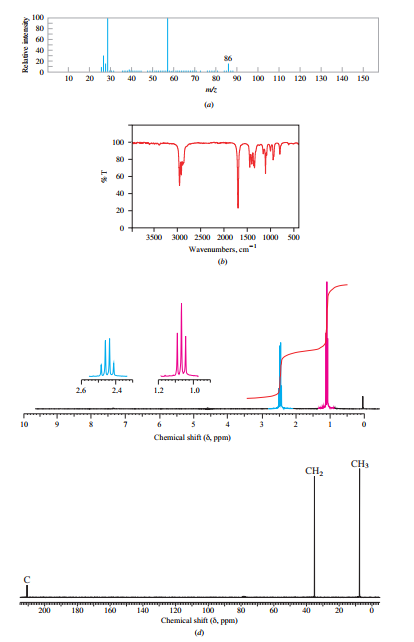
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
11. The proton NMR spectrum for a compound with formula C,HgO is shown below. The normal
carbon-13 NMR spectrum has five peaks. The infrared spectrum has a strong band at 1746 cm.
The DEPT-135 and DEPT-90 spectral results are tabulated. Draw the structure of this compound.
Normal Carbon
DEPT-135
DEPT-90
44 ppm
Negative
No peak (
125
Positive
Positive
127
Positive
Positive CA
138
No peak
No peak co
215
No peak
No peak
Proton spectrum
C,HgO
uluw
0.0
4.10
0.5
4.14
1.0
1.5
2.0
2.5
3.0
3.5
7.5
4.0
70
4.5
6.5
6.0
5.5
5.0
Which spectroscopic tool would be best for distinguishing a sample of 1,2-dichloropropane from 1,2-dibromopropane?
NMR spectroscopy
IR spectroscopy
UV/vis spectroscopy
Mass Spectrometry
How many peaks would appear in the ¹3C NMR spectrum of this compound?
Chapter 14 Solutions
Organic Chemistry - Standalone book
Ch. 14.3 - Prob. 1PCh. 14.3 - Prob. 2PCh. 14.4 - The 1H NMR signal for bromoform (CHBr3) appears at...Ch. 14.5 - identify the most shielded and least shielded...Ch. 14.5 - (a) Assign the chemical shifts 1.6, 2.2, and 4.8...Ch. 14.5 - Assign the chemical shifts 1.1, 1.7, 2.0, and 2.3...Ch. 14.5 - Assign the chemical shifts 1.6, 4.0, 7.5, 8.2, and...Ch. 14.6 - The 300-MHz 1H NMR spectrum of 1,4-dimethylbenzene...Ch. 14.6 - Prob. 9PCh. 14.6 - How many signals would you expect to find in the...
Ch. 14.7 - Describe the appearance of the 1H NMR spectrum of...Ch. 14.8 - Describe the appearance of the 1H NMR spectrum of...Ch. 14.11 - Prob. 13PCh. 14.11 - Prob. 14PCh. 14.12 - Hydrogen bonding between the oxygen of dimethyl...Ch. 14.14 - Prob. 16PCh. 14.15 - The 13C NMR spectrum of 1-bromo-3-chloropropane...Ch. 14.15 - Consider carbons x, y, and z in p-methylanisole....Ch. 14.15 - Prob. 19PCh. 14.16 - To which of the compounds of Problem 14.16 does...Ch. 14.18 - DEPT spectra for a compound with the formula...Ch. 14.20 - Vibrational frequencies are sensitive to isotopic...Ch. 14.21 - Prob. 23PCh. 14.22 - Prob. 24PCh. 14.23 - Prob. 25PCh. 14.23 - Which one of the C5H8 isomers shown has its max at...Ch. 14.24 - Knowing what to look for with respect to isotopic...Ch. 14.24 - The base peak appears at m/z105 for one of the...Ch. 14.24 - Mass spectra of 1-bromo-4-propylbenzene and...Ch. 14.25 - Prob. 30PCh. 14 - Each of the following compounds is characterized...Ch. 14 - Deduce the structure of each of the following...Ch. 14 - From among the isomeric compounds of molecular...Ch. 14 - The H1NMR spectrum of fluorene has signals at 3.8...Ch. 14 - Prob. 35PCh. 14 - H1NMR spectra of four isomeric alcohols with...Ch. 14 - Prob. 37PCh. 14 - We noted in Section 14.13 that an NMR spectrum is...Ch. 14 - Identify each of the C4H10O isomers on the basis...Ch. 14 - A compound (C3H7ClO2) exhibited three peaks in its...Ch. 14 - Label nonequivalent carbons in the following...Ch. 14 - Compounds A and B are isomers of molecular formula...Ch. 14 - C13 NMR spectra for four isomeric alkyl bromides...Ch. 14 - Prob. 44PCh. 14 - Prob. 45PCh. 14 - Identify the C3H5Br isomers on the basis of the...Ch. 14 - Prob. 47PCh. 14 - A compound (C8H10O) has the IR and H1NMR spectra...Ch. 14 - Deduce the structure of a compound having the...Ch. 14 - Figure 14.53 presents IR, H1NMR, C13NMR and mass...Ch. 14 - H1NMR, C13NMR, IR, and mass spectra are shown for...Ch. 14 - 1H NMR and IR spectra for a compound with the...Ch. 14 - FriedelCraftsalkylation of benzene with...Ch. 14 - Prob. 54DSPCh. 14 - Prob. 55DSPCh. 14 - Prob. 56DSPCh. 14 - Prob. 57DSPCh. 14 - Prob. 58DSP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Draw the correct structure from the MS, 1H NMR, 13C NMR, and IR data given below. Both of the peaks on the 1H NMR spectrum are doublets (J = 2.5 Hz) and the integration ratio is 1:1.arrow_forward'H a quartet | triplet | CDCI3 200 180 160 140 120 100 20arrow_forwardCompound CsH12 gives the following H-NMR. Draw the structure of the compound. Draw a box around the structure you want graded. 5.00 1.00 6.04 70 6.5 6.0 4.0 3.5 3.0 25 20 1.5 A student was adding bromine across the double bond of 2-butene to make 2,3-dibromobutane. After taking the NMR, the student discovered they didn't get the product expected. Based on the NMR, what product did they obtain? Draw a box around your answer. 1.00 2.01 |2.00 3.00arrow_forward
- Look at the structure of ethyl acetate in your notebook. In which region of the 13C spectrum of ethyl acetate would you NOT expect any peaks? a.0 - 50 ppm b.50 - 100 ppm c.100 - 160 ppm d.160 - 220 ppmarrow_forwardthe mass spectrum, IR and 13 C and 1 HNMR spectra for an unknown organic molecule. Determine the structure ofthe molecule.arrow_forwardQuestion 13 what compound has this given IR Spectra? 80 70 2725 2827 50 40 2976 20 10 1731 4000 2000 Wavenumbers (cm-1) O A. butyraldehyde O B. butanone O C. butanol D. butanoic acid %Transmittancearrow_forward
- Choose the best explanation for why -OH and -NH peaks are often broad singlets in a ¹H NMR spectrum. A) The greater electronegativity difference in the N-H or O-H bond compared to a C-H bond makes the peak broader. B) The greater mass involved in the N-H or O-H bond compared to a C-H bond makes the peak broader. C) The hydrogen nuclei on the carbon next to the -OH or -NH are too far away to impact the multiplicity. D) Protons in OH or NH groups are acidic enough to rapidly exchange between different molecules, so the coupling to vicinal hydrogens is not clearly observed.arrow_forwardWhat type of compound is most consistent with the IR spectrum shown below? 3000 1500 1000 4000 O None of the choices are correct. O carboxylic acid can bị nitrile (i.e., RCN) O ketone O alkene aldehyde esterarrow_forwardIn 13C NMR, carbon atoms with the lowest electron density have the largest chemical shift (largest ppm shift from 0). Identify the peaks labeled X, Y and Z in the 13C spectrum of butyl acetate as shown below. nat 200 X 150 160 140 A. X is 1; Y is 2, Z is 3 B. X is 1, Y is 3, Z is 2 C. X is 2, Y is 1, Z is 3 Enter Your Answer: 120 100 ppm OA OB OC 80 60 OD 40 20 N D. X is 2, Y is 3; Z is 1 E. X is 3, Y is 1; Z is 2 F. X is 3, Y is 2; Z is 1 OE OFarrow_forward
- Here are two chemical structures. Which technique would best distinguish between them? Br Select one: ¹H-NMR. Any of MS, IR or ¹H-NMR. Mass spectrometry. IR spectroscopy. Impossible to distinguish by any of these techniques.arrow_forwardDetermine the molecular structure of the compound in the IR spectrum data, 1H NMR ; 13C NMR and MS, explain it for each picture.arrow_forwardThe mass spectrum and 13C NMR spectrum of a hydrocarbon are shown. Propose a structure for this hydrocarbon, and explain the spectral data.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

 EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT
EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning


EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:9781305446021
Author:Lampman
Publisher:CENGAGE LEARNING - CONSIGNMENT

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning
IR Spectroscopy; Author: Professor Dave Explains;https://www.youtube.com/watch?v=_TmevMf-Zgs;License: Standard YouTube License, CC-BY