
Organic Chemistry - Standalone book
10th Edition
ISBN: 9780073511214
Author: Francis A Carey Dr., Robert M. Giuliano
Publisher: McGraw-Hill Education
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 14, Problem 41P
Label nonequivalent carbons in the following compounds.
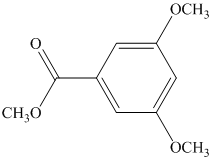
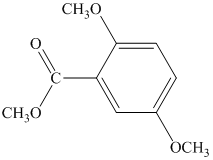
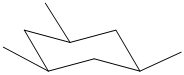
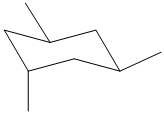
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
1. Provide proper IUPAC name to the following molecules.
5)
relationship between the pairs of structures. NOTE: Each term may be used more than
Choose the term from the five terms listed below that BEST describes the
once and not all terms need be used.
Identical
Diastereomers
Enantiomers
Constitutional isomers
Not isomers
CH3
CH3
ÇI
H3C-Br
CH3
Br
-CI
H,C.
D-H
H3C-
Br
H-
-D
Br
CH3
-CI
ČH3
OH
H3C,
CHO
OHC, OH
OH
H.
HO CHO
OHC
CH3
H.
OH
ÓH
Draw a resonance structure, complete with all formal charges and lone (unshared) electron pairs, that shows the resonance
interaction of the cyano with the ortho position in benzonitrile.
CEN
benzonitrile
• You do not have to consider stereochemistry.
• Include all valence lone pairs in your answer.
• In cases where there is more than one answer, just draw one.
Chapter 14 Solutions
Organic Chemistry - Standalone book
Ch. 14.3 - Prob. 1PCh. 14.3 - Prob. 2PCh. 14.4 - The 1H NMR signal for bromoform (CHBr3) appears at...Ch. 14.5 - identify the most shielded and least shielded...Ch. 14.5 - (a) Assign the chemical shifts 1.6, 2.2, and 4.8...Ch. 14.5 - Assign the chemical shifts 1.1, 1.7, 2.0, and 2.3...Ch. 14.5 - Assign the chemical shifts 1.6, 4.0, 7.5, 8.2, and...Ch. 14.6 - The 300-MHz 1H NMR spectrum of 1,4-dimethylbenzene...Ch. 14.6 - Prob. 9PCh. 14.6 - How many signals would you expect to find in the...
Ch. 14.7 - Describe the appearance of the 1H NMR spectrum of...Ch. 14.8 - Describe the appearance of the 1H NMR spectrum of...Ch. 14.11 - Prob. 13PCh. 14.11 - Prob. 14PCh. 14.12 - Hydrogen bonding between the oxygen of dimethyl...Ch. 14.14 - Prob. 16PCh. 14.15 - The 13C NMR spectrum of 1-bromo-3-chloropropane...Ch. 14.15 - Consider carbons x, y, and z in p-methylanisole....Ch. 14.15 - Prob. 19PCh. 14.16 - To which of the compounds of Problem 14.16 does...Ch. 14.18 - DEPT spectra for a compound with the formula...Ch. 14.20 - Vibrational frequencies are sensitive to isotopic...Ch. 14.21 - Prob. 23PCh. 14.22 - Prob. 24PCh. 14.23 - Prob. 25PCh. 14.23 - Which one of the C5H8 isomers shown has its max at...Ch. 14.24 - Knowing what to look for with respect to isotopic...Ch. 14.24 - The base peak appears at m/z105 for one of the...Ch. 14.24 - Mass spectra of 1-bromo-4-propylbenzene and...Ch. 14.25 - Prob. 30PCh. 14 - Each of the following compounds is characterized...Ch. 14 - Deduce the structure of each of the following...Ch. 14 - From among the isomeric compounds of molecular...Ch. 14 - The H1NMR spectrum of fluorene has signals at 3.8...Ch. 14 - Prob. 35PCh. 14 - H1NMR spectra of four isomeric alcohols with...Ch. 14 - Prob. 37PCh. 14 - We noted in Section 14.13 that an NMR spectrum is...Ch. 14 - Identify each of the C4H10O isomers on the basis...Ch. 14 - A compound (C3H7ClO2) exhibited three peaks in its...Ch. 14 - Label nonequivalent carbons in the following...Ch. 14 - Compounds A and B are isomers of molecular formula...Ch. 14 - C13 NMR spectra for four isomeric alkyl bromides...Ch. 14 - Prob. 44PCh. 14 - Prob. 45PCh. 14 - Identify the C3H5Br isomers on the basis of the...Ch. 14 - Prob. 47PCh. 14 - A compound (C8H10O) has the IR and H1NMR spectra...Ch. 14 - Deduce the structure of a compound having the...Ch. 14 - Figure 14.53 presents IR, H1NMR, C13NMR and mass...Ch. 14 - H1NMR, C13NMR, IR, and mass spectra are shown for...Ch. 14 - 1H NMR and IR spectra for a compound with the...Ch. 14 - FriedelCraftsalkylation of benzene with...Ch. 14 - Prob. 54DSPCh. 14 - Prob. 55DSPCh. 14 - Prob. 56DSPCh. 14 - Prob. 57DSPCh. 14 - Prob. 58DSP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- 3. Provide IUPAC systematic names for each of the following compounds. I Br Brarrow_forward[Review Topics] [References) The cyclohexane derivative shown exists primarily in the more stable of the two available chair conformations. Give the position, axial or equatorial, of each of the three groups shown in the more stable chair conformation. If a group divides its time equally between axial and equatorial positions, indicate this with ax/eq. The table of "Axial Strain Energies for Monosubstituted Cyclohexanes" found in the "Strain Energy Increments" section of the Reference tool is useful for answering this question. 6CH2CH3 Group a is Group b is Group c is Submit Answer Retry Entire Group 9 more group attempts remaining 1:06 PM 5П/2021 f12 insert prt sc delete & backspace 6 8 9.arrow_forwardThis problem is about the IUPAC (International Union of Pure and Applied Chemistry) nomenclature of organic compounds. Select the best name for the following molecule. Br F O (REI-2-bromo-4-fluorohex-3-ene O (S,Z)-2-bromo-4-fluorohex-3-ene O (R.Z)-2-bromo-4-fluorohex-3-ene O (5,D-2-bromo-4-luorohex -enearrow_forward
- The compound being investigated only has carbons and hydrogens.arrow_forwardAssign absolute configurations as R or S to each of the following compounds.arrow_forwardIs the following compound R or S. Support your claim by indicating the priority ofthe groups attached to the asymmetric carbon. H,CCH, *CH,OCH, CIarrow_forward
- How many non-equivalent carbons exist in the following compound? (Give a numberarrow_forwardHow many degrees of unsaturation does this molecule have?arrow_forwardWhat is the systematic name for the molecule depicted by the following bond line structure? Decide what the longest C-C chain is (parent) and then decide which substituents are there and how to number it to give the lowest locants. Make sure each substituent gets a locant. Separate letters from numbers by a dash, and numbers from each other by a comma. Do not put any spaces in your name.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning
07 Physical Properties of Organic Compounds; Author: Mindset;https://www.youtube.com/watch?v=UjlSgwq4w6U;License: Standard YouTube License, CC-BY