
(a)
Interpretation: The hybridization for HCN molecule needs to be determined.
Concept Introduction:
Lewis dot structure is the representation which shows the bonding between atoms present in a molecule. It shows lone pairs and bond pairs on each bonded atoms. Lewis dot structure is also known as Lewis dot formula or electron dot structure.
The sum of valence electrons must be arranged in such a way that all atoms have complete octet.
Hybridization of a molecule can be checked with the help of below formula:
(a)
Answer to Problem 19E
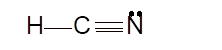
- sp hybridization
Explanation of Solution
To draw the Lewis structure, calculate the total number of valence electrons in each atom and draw the structure in such a way that each atom gets its octet configuration.
Total number of valence electrons in HCN:
Hence the best Lewis structure for HCN must be:
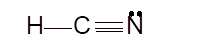
Hybridization of a molecule can be checked with the help of below formula:
(b)
Interpretation: The hybridization of
Concept Introduction:
Lewis dot structure is the representation which shows the bonding between atoms present in a molecule. It shows lone pairs and bond pairs on each bonded atoms. Lewis dot structure is also known as Lewis dot formula or electron dot structure.
The sum of valence electrons must be arranged in such a way that all atoms have complete octet.
Hybridization of a molecule can be checked with the help of below formula:
(b)
Answer to Problem 19E
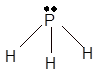
Explanation of Solution
The bond formation between the atoms takes place due to the sharing of valence electrons of bonded atoms while the remaining electrons present in outer shell represented as lone pair of electrons. To draw the Lewis structure, calculate the total number of valence electrons in each atom and draw the structure in such a way that each atom gets its octet configuration.
Total number of valence electrons in
Hence the best Lewis structure for
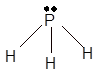
(c)
Interpretation: The hybridization of
Concept Introduction:
Lewis dot structure is the representation which shows the bonding between atoms present in a molecule. It shows lone pairs and bond pairs on each bonded atoms. Lewis dot structure is also known as Lewis dot formula or electron dot structure.
The sum of valence electrons must be arranged in such a way that all atoms have complete octet.
Hybridization of a molecule can be checked with the help of below formula:
(c)
Answer to Problem 19E
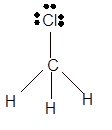
Explanation of Solution
The bond formation between the atoms takes place due to the sharing of valence electrons of bonded atoms while the remaining electrons present in outer shell represented as lone pair of electrons. To draw the Lewis structure, calculate the total number of valence electrons in each atom and draw the structure in such a way that each atom gets its octet configuration.
Total number of valence electrons in
Hence the best Lewis structure for
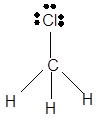
(d)
Interpretation: The hybridization of
Concept Introduction:
Lewis dot structure is the representation which shows the bonding between atoms present in a molecule. It shows lone pairs and bond pairs that existing on each bonded atom. Lewis dot structure is also known as Lewis dot formula or electron dot structure.
The sum of valence electrons must be arranged in such a way that all atoms must get octet configuration (8 electrons).
Hybridization of a molecule can be checked with the help of below formula:
(d)
Answer to Problem 19E
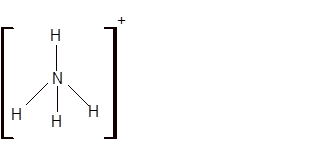
Explanation of Solution
The bond formation between the atoms takes place due to the sharing of valence electrons of bonded atoms while the remaining electrons present in outer shell represented as lone pair of electrons. To draw the Lewis structure, calculate the total number of valence electrons in each atom and draw the structure in such a way that each atom gets its octet configuration.
Total number of valence electrons in
Hence the best Lewis structure for
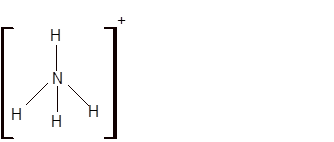
(e)
Interpretation: The hybridization of
Concept Introduction:
Lewis dot structure is the representation which shows the bonding between atoms present in a molecule. It shows lone pairs and bond pairs on each bonded atoms. Lewis dot structure is also known as Lewis dot formula or electron dot structure.
The sum of valence electrons must be arranged in such a way that all atoms have complete octet.
Hybridization of a molecule can be checked with the help of below formula:
(e)
Answer to Problem 19E
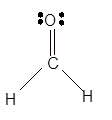
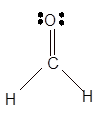
Explanation of Solution
The bond formation between the atoms takes place due to the sharing of valence electrons of bonded atoms while the remaining electrons present in outer shell represented as lone pair of electrons. To draw the Lewis structure, calculate the total number of valence electrons in each atom and draw the structure in such a way that each atom gets its octet configuration.
Total number of valence electrons in
Hence the best Lewis structure for
(f)
Interpretation: The hybridization of
Concept Introduction:
Lewis dot structure is the representation which shows the bonding between atoms present in a molecule. It shows lone pairs and bond pairs that existing on each bonded atom. Lewis dot structure is also known as Lewis dot formula or electron dot structure.
The sum of valence electrons must be arranged in such a way that all atoms must get octet configuration (8 electrons).
Hybridization of a molecule can be checked with the help of below formula:
(f)
Answer to Problem 19E
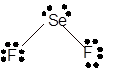
Explanation of Solution
The bond formation between the atoms takes place due to the sharing of valence electrons of bonded atoms while the remaining electrons present in outer shell represented as lone pair of electrons. To draw the Lewis structure, calculate the total number of valence electrons in each atom and draw the structure in such a way that each atom gets its octet configuration.
Total number of valence electrons in
Hence, the best Lewis structure for
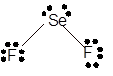
(g)
Interpretation: The hybridization of
Concept Introduction:
Lewis dot structure is the representation which shows the bonding between atoms present in a molecule. It shows lone pairs and bond pairs that existing on each bonded atom. Lewis dot structure is also known as Lewis dot formula or electron dot structure.
The sum of valence electrons must be arranged in such a way that all atoms must get octet configuration (8 electrons).
Hybridization of a molecule can be checked with the help of below formula:
(g)
Answer to Problem 19E
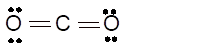
sp- hybridization
Explanation of Solution
The bond formation between the atoms takes place due to the sharing of valence electrons of bonded atoms while the remaining electrons present in outer shell represented as lone pair of electrons. To draw the Lewis structure, calculate the total number of valence electrons in each atom and draw the structure in such a way that each atom gets its octet configuration.
Total number of valence electrons in
Hence the best Lewis structure for
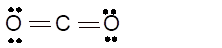
(h)
Interpretation: The hybridization of
Concept Introduction:
Lewis dot structure is the representation which shows the bonding between atoms present in a molecule. It shows lone pairs and bond pairs that existing on each bonded atom. Lewis dot structure is also known as Lewis dot formula or electron dot structure.
The sum of valence electrons must be arranged in such a way that all atoms must get octet configuration (8 electrons).
Hybridization of a molecule can be checked with the help of below formula:
(h)
Answer to Problem 19E

Explanation of Solution
The bond formation between the atoms takes place due to the sharing of valence electrons of bonded atoms while the remaining electrons present in outer shell represented as lone pair of electrons. To draw the Lewis structure, calculate the total number of valence electrons in each atom and draw the structure in such a way that each atom gets its octet configuration.
Total number of valence electrons in
Hence the best Lewis structure for

(i)
Interpretation: The hybridization of HBr needs to be determined.
Concept Introduction:
Lewis dot structure is the representation which shows the bonding between atoms present in a molecule. It shows lone pairs and bond pairs on each bonded atoms. Lewis dot structure is also known as Lewis dot formula or electron dot structure.
The sum of valence electrons must be arranged in such a way that all atoms have complete octet.
Hybridization of a molecule can be checked with the help of below formula:
(i)
Answer to Problem 19E

Explanation of Solution
The bond formation between the atoms takes place due to the sharing of valence electrons of bonded atoms while the remaining electrons present in outer shell represented as lone pair of electrons. To draw the Lewis structure, calculate the total number of valence electrons in each atom and draw the structure in such a way that each atom gets its octet configuration.
Total number of valence electrons in
Hence the best Lewis structure for

(j)
Interpretation: The hybridization of
Concept Introduction:
Lewis dot structure is the representation which shows the bonding between atoms present in a molecule. It shows lone pairs and bond pairs that existing on each bonded atom. Lewis dot structure is also known as Lewis dot formula or electron dot structure.
The sum of valence electrons must be arranged in such a way that all atoms must get octet configuration (8 electrons).
Hybridization of a molecule can be checked with the help of below formula:
(j)
Answer to Problem 19E
Explanation of Solution
To draw the Lewis structure, calculate the total number of valence electrons in each atom and draw the structure in such a way that each atom gets its octet configuration.
Total number of valence electrons in
Hence the best Lewis structure for
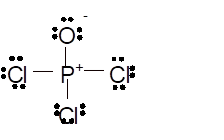
Total number of valence electrons in
Hence the best Lewis structure for
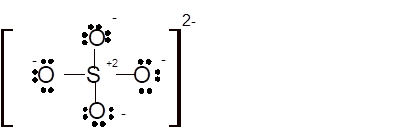
Total number of valence electrons in
Hence the best Lewis structure for
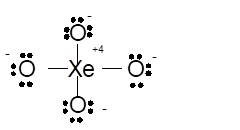
Total number of valence electrons in
Hence the best Lewis structure for
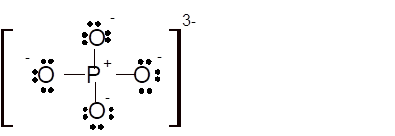
Total number of valence electrons in
Hence the best Lewis structure for
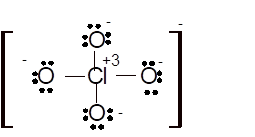
(k)
Interpretation: The hybridization of
Concept Introduction:
Lewis dot structure is the representation which shows the bonding between atoms present in a molecule. It shows lone pairs and bond pairs that existing on each bonded atom. Lewis dot structure is also known as Lewis dot formula or electron dot structure.
The sum of valence electrons must be arranged in such a way that all atoms must get octet configuration (8 electrons).
Hybridization of a molecule can be checked with the help of below formula:
(k)
Answer to Problem 19E
Explanation of Solution
The bond formation between the atoms takes place due to the sharing of valence electrons of bonded atoms while the remaining electrons present in outer shell represented as lone pair of electrons. To draw the Lewis structure, calculate the total number of valence electrons in each atom and draw the structure in such a way that each atom gets its octet configuration.
Total number of valence electrons in
Hence the best Lewis structure for
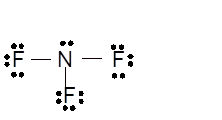
Total number of valence electrons in
Hence the best Lewis structure for
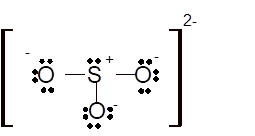
Total number of valence electrons in
Hence the best Lewis structure for
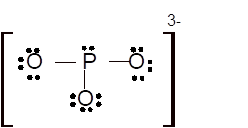
Total number of valence electrons in
Hence the best Lewis structure for
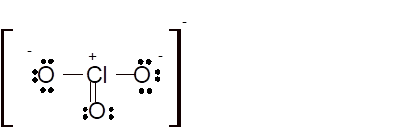
(l)
Interpretation: The hybridization of
Concept Introduction:
Lewis dot structure is the representation which shows the bonding between atoms present in a molecule. It shows lone pairs and bond pairs that existing on each bonded atom. Lewis dot structure is also known as Lewis dot formula or electron dot structure.
The sum of valence electrons must be arranged in such a way that all atoms must get octet configuration (8 electrons).
Hybridization of a molecule can be checked with the help of below formula:
(l)
Answer to Problem 19E
Explanation of Solution
The bond formation between the atoms takes place due to the sharing of valence electrons of bonded atoms while the remaining electrons present in outer shell represented as lone pair of electrons. To draw the Lewis structure, calculate the total number of valence electrons in each atom and draw the structure in such a way that each atom gets its octet configuration.
Total number of valence electrons in
Hence the best Lewis structure for
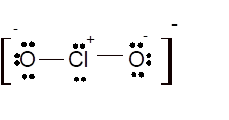
Total number of valence electrons in
Hence the best Lewis structure for
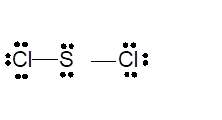
Total number of valence electrons in
Hence the best Lewis structure for
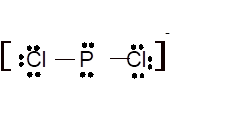
(m)
Interpretation: The hybridization of
Concept Introduction:
Lewis dot structure is the representation which shows the bonding between atoms present in a molecule. It shows lone pairs and bond pairs present on bonded atom. Lewis dot structure is also known as Lewis dot formula or electron dot structure.
The sum of valence electrons must be arranged in such a way that all atoms must get octet configuration (8 electrons).
Hybridization of a molecule can be checked with the help of below formula:
(m)
Answer to Problem 19E
Explanation of Solution
The bond formation between the atoms takes place due to the sharing of valence electrons of bonded atoms while the remaining electrons present in outer shell represented as lone pair of electrons. To draw the Lewis structure, calculate the total number of valence electrons in each atom and draw the structure in such a way that each atom gets its octet configuration.
According to the given molecular formula, the structure of
Hence the best Lewis structure for

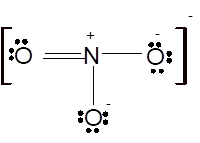
According to the given molecular formula, the structure of
Hence the best Lewis structure for
According to the given molecular formula, the structure of
Hence the best Lewis structure for
(n)
Interpretation: The hybridization of
Concept Introduction:
Lewis dot structure is the representation which shows the bonding between atoms present in a molecule. It shows lone pairs and bond pairs that existing on present on each bonded atom. Lewis dot structure is also known as Lewis dot formula or electron dot structure.
The sum of valence electrons must be arranged in such a way that all atoms must get octet configuration (8 electrons).
Hybridization of a molecule can be checked with the help of below formula:
(n)
Answer to Problem 19E
Explanation of Solution
The bond formation between the atoms takes place due to the sharing of valence electrons of bonded atoms while the remaining electrons present in outer shell represented as lone pair of electrons. To draw the Lewis structure, calculate the total number of valence electrons in each atom and draw the structure in such a way that each atom gets its octet configuration.
According to the given molecular formula, the structure of
Hence the best Lewis structure for
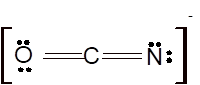
According to the given molecular formula, the structure of
Hence the best Lewis structure for
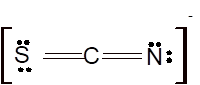
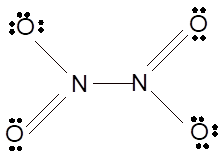
According to the given molecular formula, the structure of
Hence the best Lewis structure for

Want to see more full solutions like this?
Chapter 14 Solutions
Chemical Principles
- Give the expected hybridization of the central atom for the molecules in Exercises 21 and 22.arrow_forwardGive the Lewis structure, molecular structure, and hybridization of the oxygen atom for OF2. Would you expect OF2 to be a strong oxidizing agent like O2F2 discussed in Exercise 61?arrow_forwardWhat hybrid orbitals would be expected for the central atom in each of the following molecules or ions?arrow_forward
- Acrylonitrile, C3H3N is the building mer Orlon. Its Lewis structure is What is the hybridization of nitrogen and of the three numbered carbon atoms?arrow_forwardIn the hybrid orbital model, compare and contrast bonds with bonds. What orbitals form the bonds and what orbitals form the bonds? Assume the z-axis is the internuclear axis.arrow_forwardWhy do we hybtidize atomic orbitals to explain the bonding in covalent compounds? What type of bonds form from hybrid orbitals: sigma or pi? Explain.arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
 Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning





