
Interpretation: To identify the structure with at least one double bond.
Concept Introduction: The compound with double bond can be determined by drawing the Lewis structure of each compound. When the Lewis structure for each compound is given, the number of bonds between the central metal atom and the neighboring atoms can be determined including the lone pair of atoms. The number of valence electrons of both the central metal atom and the surrounding atom will determine the number of bonds that can be formed.
Answer to Problem 112A
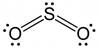
Explanation of Solution
The number of valence electrons in the sulfur atom is 6 and the number of valence electrons in oxygen is also 6. Sulfur is considered as the central metal atom and the oxygen will be surrounding the central metal atom. Sulfur has the tendency to form 6 bonds while oxygen has a tendency to form only two bonds. Therefore, the sulfur is attached to both the oxygens with a double bond and contain a lone pair of electrons.
In hydrogen sulfide, even though sulfur has the ability to form a double bond, hydrogen cannot form a double bond. Similarly, in Phosphorous trichloride, phosphorous has the ability to form a double bond but chlorine cannot form a double bond. Therefore, in both the structures there is no possibility of a double bond.
Chapter 14 Solutions
Chemistry 2012 Student Edition (hard Cover) Grade 11
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





