
Fundamentals of General, Organic, and Biological Chemistry (8th Edition)
8th Edition
ISBN: 9780134015187
Author: John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. Peterson
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Question
Chapter 13.5, Problem 13.3CIAP
Interpretation Introduction
Interpretation:
The colour absorbed by the Tetrabromofluorescein dye has to be predicted.
Concept Introduction:
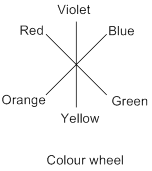
Colour wheel: The arrangement of rainbow colours called ‘VIBGYOR’ around a circle; the interchanging and mixing of primary (red, blue, and yellow) colours between them results in secondary, tertiary colours etc.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Biochemistry
What is the process of "transamination" in either the muscles or the liver, that involves keto acid or glutamic acid?
Please explain how the steps work. Thank you!
Biochemistry
Please help. Thank you
What is the importance of glutamic acid in the metabolism of nitrogen from amino acids? (we know therole; it’s used to remove the nitrogen from amino acids so that the remaining carbon skeleton can bebroken down by the “usual” pathways, but what is the important, unique role that only glutamicacid/glutamate can do?)
Biochemistry
Please help. Thank you
When carbamyl phosphate is joined to L-ornathine, where does the energy for the reaction come from?
Chapter 13 Solutions
Fundamentals of General, Organic, and Biological Chemistry (8th Edition)
Ch. 13.2 - Prob. 13.1PCh. 13.2 - Prob. 13.2PCh. 13.2 - What are the IUPAC names of the two alkenes shown...Ch. 13.3 - Prob. 13.4PCh. 13.3 - Prob. 13.5PCh. 13.3 - Prob. 13.6KCPCh. 13.5 - Prob. 13.7PCh. 13.5 - Many biological transformations can be simply...Ch. 13.5 - (a) After the reaction of 11-cis-retinal with...Ch. 13.5 - Prob. 13.2CIAP
Ch. 13.5 - Prob. 13.3CIAPCh. 13.6 - Prob. 13.9PCh. 13.6 - Prob. 13.10PCh. 13.6 - Prob. 13.11PCh. 13.6 - Prob. 13.12PCh. 13.6 - Prob. 13.13PCh. 13.6 - Prob. 13.14KCPCh. 13.6 - Prob. 13.1MRPCh. 13.6 - Prob. 13.2MRPCh. 13.6 - Prob. 13.3MRPCh. 13.7 - Prob. 13.15PCh. 13.7 - Prob. 13.16PCh. 13.8 - Prob. 13.17PCh. 13.8 - Polychlorotrifluoroethylene (PCTFE (Kel-F)) is a...Ch. 13.9 - Prob. 13.19PCh. 13.9 - Prob. 13.20PCh. 13.9 - Prob. 13.21KCPCh. 13.10 - What products will be formed when toluene is...Ch. 13.10 - Prob. 13.23PCh. 13.10 - Prob. 13.4CIAPCh. 13.10 - Prob. 13.5CIAPCh. 13.10 - Prob. 13.6CIAPCh. 13 - Prob. 13.24UKCCh. 13 - Prob. 13.25UKCCh. 13 - Prob. 13.26UKCCh. 13 - Draw the product from reaction of the following...Ch. 13 - Prob. 13.28UKCCh. 13 - Prob. 13.29UKCCh. 13 - Prob. 13.30APCh. 13 - Prob. 13.31APCh. 13 - What family-name endings are used for alkenes,...Ch. 13 - Prob. 13.33APCh. 13 - Prob. 13.34APCh. 13 - Write structural formulas for compounds that meet...Ch. 13 - Prob. 13.36APCh. 13 - Prob. 13.37APCh. 13 - Draw structures corresponding to the following...Ch. 13 - Draw structures corresponding to the following...Ch. 13 - Seven alkynes have the formula C6H10. Draw them,...Ch. 13 - Prob. 13.41APCh. 13 - Prob. 13.42APCh. 13 - There are four different pentenes having the...Ch. 13 - Prob. 13.44APCh. 13 - Prob. 13.45APCh. 13 - Draw line structures for the following alkenes....Ch. 13 - Which compound(s) in Problem 13.43 can exist as...Ch. 13 - Prob. 13.48APCh. 13 - Prob. 13.49APCh. 13 - Which of the following pairs are isomers, and...Ch. 13 - Prob. 13.51APCh. 13 - Prob. 13.52APCh. 13 - Prob. 13.53APCh. 13 - Prob. 13.54APCh. 13 - Prob. 13.55APCh. 13 - Prob. 13.56APCh. 13 - Prob. 13.57APCh. 13 - Prob. 13.58APCh. 13 - Prob. 13.59APCh. 13 - What alkene could you use to make the following...Ch. 13 - Prob. 13.61APCh. 13 - Prob. 13.62APCh. 13 - Prob. 13.63APCh. 13 - Prob. 13.64APCh. 13 - For each of the following reagents, decide whether...Ch. 13 - Prob. 13.66APCh. 13 - Prob. 13.67APCh. 13 - Prob. 13.68APCh. 13 - Salicylic acid (o-hydroxybenzoic acid) is used as...Ch. 13 - The following names are incorrect by IUPAC rules....Ch. 13 - Prob. 13.71CPCh. 13 - Prob. 13.72CPCh. 13 - Prob. 13.73CPCh. 13 - Prob. 13.74CPCh. 13 - Menthene, a compound found in mint plants, has the...Ch. 13 - Prob. 13.76CPCh. 13 - Prob. 13.77CPCh. 13 - Two products are possible when 2-pentene is...Ch. 13 - Ocimene, a compound isolated from the herb basil,...Ch. 13 - Describe how you could prepare the following...Ch. 13 - Which of the following compounds are capable of...Ch. 13 - Prob. 13.82GPCh. 13 - Superglue is an alkene polymer made from the...Ch. 13 - Draw all possible C5H10 alkene isomers having a...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biochemistry and related others by exploring similar questions and additional content below.Similar questions
- Biochemistry Question Please help. Thank you What is the function of glutamate dehydrogenase?arrow_forwardBiochemistry Question Please help. Thank you How and why does a high protein diet affect the enzymes of the urea cycle?arrow_forwardBiochemistry What is the importance of the glucose-alanine cycle?arrow_forward
- Biochemistry Assuming 2.5 molecules of ATP per oxidation of NADH/(H+) and 1.5molecules of ATP per oxidation of FADH2, how many ATP are produced per molecule of pyruvate? Please help. Thank youarrow_forward1. How would you explain the term ‘good food’? 2. How would you define Nutrition? 3. Nutrients are generally categorised into two forms. Discuss.arrow_forwardBiochemistry Question. Please help solve. Thank you! Based upon knowledge of oxidation of bioorganic compounds and howmuch energy is released during their oxidation, rank the following, from most to least, with respect to how much energy would be produced from each during their oxidation. Explain your placement for each one.arrow_forward
- Biochemistry Question.For the metabolism of amino acids what is the first step for theirbreakdown? Why is it necessary for this breakdown product to be transported to the liver? For the catabolism of the carbon backbone of these amino acids, there are 7 entry points into the “standard” metabolic pathways. List these 7 entry points and which amino acids are metabolized to these entry points. Please help. Thank you!arrow_forwardBiochemistry Question. Please help. Thank you. You are studying pyruvate utilization in mammals for ATP production under aerobic conditions and have synthesized pyruvate with Carbon #1 labelled with radioactive C14. After only one complete cycle of the TCA cycle, which of the TCA cycle intermediates would be labeled with C14? Explain your answer. Interestingly, you find C14 being excreted in the urine. How does it get there?arrow_forwardBiochemistry question. Please help with. Thanks in advance For each of the enzymes listed below, explain what the enzyme does including function, names (or structures) of the substrate and products and the pathway(s) (if applicable) it is/are found in. (a) ATP synthetase (b) succinate dehydrogenase (c) isocitrate lyase (d) acetyl CoA carboxylase (e) isocitrate dehydrogenase (f) malate dehydrogenasearrow_forward
- Draw and name each alcohol and classify it as primary, secondary, or tertiary. Explain your answer thoroughly.arrow_forwardDraw the product of each reaction. If there are multiple products, draw only the major product. Explain your answer thoroughly.arrow_forwardIdentify the type of bond in the following disaccharides. Number your carbons to show work. Explain your answer thoroughly. Draw the number of carbons also.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Principles Of Radiographic Imaging: An Art And A ...Health & NutritionISBN:9781337711067Author:Richard R. Carlton, Arlene M. Adler, Vesna BalacPublisher:Cengage LearningEssentials of Pharmacology for Health ProfessionsNursingISBN:9781305441620Author:WOODROWPublisher:Cengage
Principles Of Radiographic Imaging: An Art And A ...Health & NutritionISBN:9781337711067Author:Richard R. Carlton, Arlene M. Adler, Vesna BalacPublisher:Cengage LearningEssentials of Pharmacology for Health ProfessionsNursingISBN:9781305441620Author:WOODROWPublisher:Cengage


Principles Of Radiographic Imaging: An Art And A ...
Health & Nutrition
ISBN:9781337711067
Author:Richard R. Carlton, Arlene M. Adler, Vesna Balac
Publisher:Cengage Learning

Essentials of Pharmacology for Health Professions
Nursing
ISBN:9781305441620
Author:WOODROW
Publisher:Cengage
GCSE Chemistry - Acids and Bases #34; Author: Cognito;https://www.youtube.com/watch?v=vt8fB3MFzLk;License: Standard youtube license