
(a)
Interpretation:
Three possible structures of given
Concept Introduction:
Line (skeletal) structure: In organic compounds, the series of atoms in the compound are bonded together which shown by drawing a line between them. End of line segment represents carbon.
Structural Description of Alkene: An alkene contains
(b)
Interpretation:
Three possible structures of given
Concept Introduction:
Line (skeletal) structure: In organic compounds, the series of atoms in the compound are bonded together which shown by drawing a line between them. End of line segment represents carbon.
Structural Description of Alkyne: The general formula of alkyne is
(c)
Interpretation:
Structure of given monosubstituted benzene has to be drawn.
Concept Introduction:
Line (skeletal) structure: In organic compounds, the series of atoms in the compound are bonded together which shown by drawing a line between them. End of line segment represents carbon.
Structural Description of monosubstituted benzene: Monosubstituted benzenes are named using ‘-benzene’ as the parent name. The position of one substituent can be located at any one carbon of the benzene ring.
(d)
Interpretation:
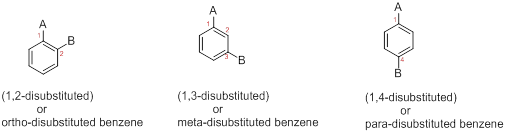
Three possible structures of given disubstituted benzene has to be drawn.
Concept Introduction:
Line (skeletal) structure: In organic compounds, the series of atoms in the compound are bonded together which shown by drawing a line between them. End of line segment represents carbon.
Structural Description of Disubstituted benzene: When benzene has more than one substituent, the position of those substituents is indicated by numbers as (1,2), (1,3), and (1,4) or by (1,2-disubstitued) as ortho(o-),(1,3-disubstitued) as meta(m-) or(1,4-disubstitued) as para(p-) in prefix.
Want to see the full answer?
Check out a sample textbook solution
Chapter 13 Solutions
Fundamentals of General, Organic, and Biological Chemistry (8th Edition)
- Draw structural formulas for all of the following. Q.) Alcohols with the molecular formula C4H10Oarrow_forwardWrite chemical names for the following compounds: (a) Thymidine (b) Cytosine (c) Uracil (d) Xanthine (e) Guanine () 2,4-dioxy-6-carboxy pyrimidine (g) CAMP (h) dTTParrow_forwardWrite the IUPAC name for each unsaturated hydrocarbon. (a) CH,=CH(CH,),CH3 CH3 CH3 H,C- (b) CH3 (c) CH3 alaTTarrow_forward
- Compound A undergoes a reaction with hydrogen bromide, HBr to produce2-bromobutane. A exists as cis-trans isomers and decolourises brominesolution in methylene chloride, CH2Cl2. a)Draw and name the structure of compound D. b)Draw two (2) constitutional isomers of compound Darrow_forwardDraw structures of the following compounds:(a) cis-Hept-3-ene(b) cis-4-Methylpent-2-ene(c) trans-2,5-Dimethylhex-3-enearrow_forwardAre the following molecules chiral or achiral? If they are chiral, identify the chiral carbon atom(s).(a) Pentan-3-ol (b) 2-Bromobutane(c) 2-Methylcyclohexanolarrow_forward
- Identify the acid on the left and its conjugate base on the right in the following equations:(a) HOCl + H2O ↔ H3O+ + OCl-(b) HONH2 + H2O ↔ HONH3+ + OH-(c) NH4+ + H2O ↔ NH3 + H3O+(d) 2HCO3-2 ↔ H2CO3 + CO3-2 (e) PO4-3 + H2PO4- ↔ 2HPO4-2arrow_forwardDraw a Lewis structure for hydroxylamine, NH2OH.arrow_forwarda) what two types of bonds are present in alkenes but not alkanes? b) draw the structure of an alkene that would not have one of the two bondsarrow_forward
- Draw structures corresponding to the following names:(a) 2,4-Dimethylheptan-2-ol(b) 2,2-Diethylcyclohexanol(c) 5-Ethyl-5-methylheptan-1-ol(d) 4-Ethylhexan-2-ol(e) 3-Methoxycyclooctanol(f) 3,3-Dimethylheptane-1,6-diolarrow_forwardDefine the following terms:(a) Chiral (b) Achiral(c) Chiral carbon (d) Enantiomerarrow_forwardDraw condensed structural formulas for the two carboxylic acids with the molecular formula C4H8O2arrow_forward
 BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman
BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman
Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY
Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON
Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON





