
Fundamentals of General, Organic, and Biological Chemistry (8th Edition)
8th Edition
ISBN: 9780134015187
Author: John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. Peterson
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 13.5, Problem 13.8P
Many biological transformations can be simply classified as additions, eliminations, or substitutions. How would you classify the following reactions?
(a) Fumaric acid to malic acid (found in the citric acid cycle. Section 21.8)
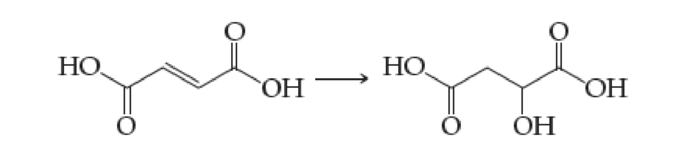
(b) 2-Phosphoglyceric acid to phosphoenolpyruvic acid (found in glycolysis, Section 22.3)
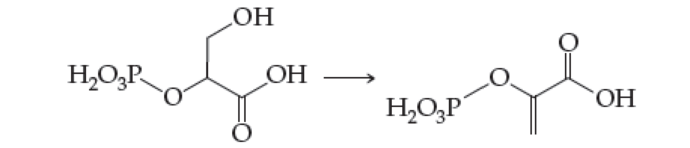
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
a)
2-Hydroxy-carboxylic acids in the citric acid cycle and in glycolysis (3 answers):
b) how do the ring systems in tryptophan (indole ring) and in histidine (imidazole ring) fulfill the Hückel rule?
c) How does the aromatic state affect the basicity of the ring nitrogens?
(a) Convert the ball-and-stick model of B, one of the eight synthetic intermediates in the citric acid cycle, to a structural formula, and name the compound. (b) What is the immediate precursor of B in the citric acid cycle; that is, what compound forms B as a reaction product? (c) What compound is formed from B in the citric acid cycle?
(a) Write a balanced equation for the conversion of aspartate into glucose through the intermediate oxaloacetate. Which coenzymes participate in this transformation? (b) Write a balanced equation for the conversion of aspartate into oxaloacetate through the intermediate fumarate
Chapter 13 Solutions
Fundamentals of General, Organic, and Biological Chemistry (8th Edition)
Ch. 13.2 - Prob. 13.1PCh. 13.2 - Prob. 13.2PCh. 13.2 - What are the IUPAC names of the two alkenes shown...Ch. 13.3 - Prob. 13.4PCh. 13.3 - Prob. 13.5PCh. 13.3 - Prob. 13.6KCPCh. 13.5 - Prob. 13.7PCh. 13.5 - Many biological transformations can be simply...Ch. 13.5 - (a) After the reaction of 11-cis-retinal with...Ch. 13.5 - Prob. 13.2CIAP
Ch. 13.5 - Prob. 13.3CIAPCh. 13.6 - Prob. 13.9PCh. 13.6 - Prob. 13.10PCh. 13.6 - Prob. 13.11PCh. 13.6 - Prob. 13.12PCh. 13.6 - Prob. 13.13PCh. 13.6 - Prob. 13.14KCPCh. 13.6 - Prob. 13.1MRPCh. 13.6 - Prob. 13.2MRPCh. 13.6 - Prob. 13.3MRPCh. 13.7 - Prob. 13.15PCh. 13.7 - Prob. 13.16PCh. 13.8 - Prob. 13.17PCh. 13.8 - Polychlorotrifluoroethylene (PCTFE (Kel-F)) is a...Ch. 13.9 - Prob. 13.19PCh. 13.9 - Prob. 13.20PCh. 13.9 - Prob. 13.21KCPCh. 13.10 - What products will be formed when toluene is...Ch. 13.10 - Prob. 13.23PCh. 13.10 - Prob. 13.4CIAPCh. 13.10 - Prob. 13.5CIAPCh. 13.10 - Prob. 13.6CIAPCh. 13 - Prob. 13.24UKCCh. 13 - Prob. 13.25UKCCh. 13 - Prob. 13.26UKCCh. 13 - Draw the product from reaction of the following...Ch. 13 - Prob. 13.28UKCCh. 13 - Prob. 13.29UKCCh. 13 - Prob. 13.30APCh. 13 - Prob. 13.31APCh. 13 - What family-name endings are used for alkenes,...Ch. 13 - Prob. 13.33APCh. 13 - Prob. 13.34APCh. 13 - Write structural formulas for compounds that meet...Ch. 13 - Prob. 13.36APCh. 13 - Prob. 13.37APCh. 13 - Draw structures corresponding to the following...Ch. 13 - Draw structures corresponding to the following...Ch. 13 - Seven alkynes have the formula C6H10. Draw them,...Ch. 13 - Prob. 13.41APCh. 13 - Prob. 13.42APCh. 13 - There are four different pentenes having the...Ch. 13 - Prob. 13.44APCh. 13 - Prob. 13.45APCh. 13 - Draw line structures for the following alkenes....Ch. 13 - Which compound(s) in Problem 13.43 can exist as...Ch. 13 - Prob. 13.48APCh. 13 - Prob. 13.49APCh. 13 - Which of the following pairs are isomers, and...Ch. 13 - Prob. 13.51APCh. 13 - Prob. 13.52APCh. 13 - Prob. 13.53APCh. 13 - Prob. 13.54APCh. 13 - Prob. 13.55APCh. 13 - Prob. 13.56APCh. 13 - Prob. 13.57APCh. 13 - Prob. 13.58APCh. 13 - Prob. 13.59APCh. 13 - What alkene could you use to make the following...Ch. 13 - Prob. 13.61APCh. 13 - Prob. 13.62APCh. 13 - Prob. 13.63APCh. 13 - Prob. 13.64APCh. 13 - For each of the following reagents, decide whether...Ch. 13 - Prob. 13.66APCh. 13 - Prob. 13.67APCh. 13 - Prob. 13.68APCh. 13 - Salicylic acid (o-hydroxybenzoic acid) is used as...Ch. 13 - The following names are incorrect by IUPAC rules....Ch. 13 - Prob. 13.71CPCh. 13 - Prob. 13.72CPCh. 13 - Prob. 13.73CPCh. 13 - Prob. 13.74CPCh. 13 - Menthene, a compound found in mint plants, has the...Ch. 13 - Prob. 13.76CPCh. 13 - Prob. 13.77CPCh. 13 - Two products are possible when 2-pentene is...Ch. 13 - Ocimene, a compound isolated from the herb basil,...Ch. 13 - Describe how you could prepare the following...Ch. 13 - Which of the following compounds are capable of...Ch. 13 - Prob. 13.82GPCh. 13 - Superglue is an alkene polymer made from the...Ch. 13 - Draw all possible C5H10 alkene isomers having a...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biochemistry and related others by exploring similar questions and additional content below.Similar questions
- a.) in human, under what condition will pyruvate be converted to lactate? what type of reaction occurs when pyruvate is converted to lactate? b.) in human, what happens to pyruvate when there is sufficient oxygen supply? which enzyme is involved in this chemical reaction?arrow_forwardWhich of the following statements about isozymes is correct? (Select all that apply.) (a) Isozymes allow reactions to be optimized under different conditions. (b) The main reason for isozymes is so that a given reaction is never completely inhibited. (c) Isozymes have completely different active sites. (d) Bind the same substrates but form different products. (e) Isozymes display different physical properties, such as kinetic parameters.arrow_forwardWhile fatty acids longer than 20 carbons are rarely found in foods, lignoceric acid (24:0) is found in a variety of tree nuts. Answer the following based on the conversion of a molecule of lignoceric acid to 8-hydroxybutyrate. (a) What are the 8-oxidation products and how many ATP are required during activation for one molecule of lignoceric acid? (b) Given the following, how many molecules of 8-hydroxybutyrate can be produced? CoA 2 2 CoA NADH NAD+ H+ OH ẞ-hydroxybutyrate (c) Based on the total NADH and FADH2 available after converting lignoceric acid into 8-hydroxybutyrate, what is the maximum yield of ATP that can be produced in the liver? Don't forget to include any ATP required for activation steps.arrow_forward
- Which of the following is a true statement regarding sphingolipid synthesis? (A) The first step in sphingolipid synthesis is the condensation of palmitoyl CoA with aspartate to form b-ketosphinganine.(B) This process requires the reduction of a ketone that uses NADH as the reducing agent.(C) A fatty acid is attached to dihydrosphingosine to form dihydroceramide. (D) FAD is using as an oxidizing agent to remove a double bond from dihydroceramide.(E) The formation of sphingomyelin requires the attachment of a glucose or galactose molecule to ceramide.arrow_forwardThe phosphorylation of glucose to glucose 1-phosphate requires 5.0 kcal/mol of energy. This unfavorable reaction can be driven by the hydrolysis of ATP to ADP. (a) Write the equation for the coupled reaction. (b) How much energy is released in the coupled reaction? glucose + HPO4---------->2– glucose 1-phosphate + H2Oarrow_forward15.12) Determine whether each of the following metabolic processes occurs in the cytoplasm (outside of the mitochondria) or in the mitochondria. a) glycolysis cytoplasm b) citric acid cycle mitochondria c) beta oxidation mitochondria d) oxidative phosphorylation mitochondria EXPLANATION: The enzymes that catalyze the reactions of the citric acid cycle, beta-oxidation, and oxidative phosphorylation are all located in the mitochondria.arrow_forward
- Shown below are reduction potentials for four half-reactions. Which of the coupled reactions is favorable? (Note that for Cytochrome c you must multiply the reduction potential by 2 for each coupled reaction because only one electron is involved) a) 2 Cytochrome c (Fe3+) + H2O ó 2 Cytochrome c (Fe2+) + O2 b) NADH + Succinate- ó NAD+ + Fumarate- c) Fumarate- + H2O ó Succinate- + O2 d) All of the abovearrow_forwardEach of the following enzymes catalyzes what sort of reaction?citrate decarboxylase (a. citrate decarboxylase) (b. citrate decarboxyoxalate reductase (b)c. transaminase of serinearrow_forwardMost glucose in the human diet comes from plants. Unlike humans, plants transport sucrose instead of glucose. The conversion of sucrose to glucose and fructose is a reversible reaction as shown in Reaction 25.1: Reaction 25.1 Sucrose (aq) + H2O (l) ⇄ Glucose (aq) + Fructose (aq) Under conditions typical of human metabolism, the Keq for Reaction 25.1 is 140,000. At equilibrium, would you expect more of the reactant sucrose or more of the products glucose and fructose to be present? Explain reasoning.arrow_forward
- The citric acid cycle contains four 4-carbon dicarboxylic acids. (a) Name them.(b) Arrange them in order from least oxidized to mostoxidized.arrow_forwardThis is a conjectural question: If the reactive part of coenzyme A is the thioester, why is the molecule socomplicated?arrow_forwardUsing the glycerol-3-phosphate shuttle, determine how many ATP can be produced from one mole of each of the following compounds on complete oxidation? a.) Mannose b.) Dihydroxyacetone phosphate c.) Citrate d.) Malate e.) Succinate Answers vary between 32 moles, 32 moles, 16 moles, 10 moles, 4 moles, and 2.5 molesarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman
BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman
Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY
Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON
Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON

Biochemistry
Biochemistry
ISBN:9781319114671
Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.
Publisher:W. H. Freeman

Lehninger Principles of Biochemistry
Biochemistry
ISBN:9781464126116
Author:David L. Nelson, Michael M. Cox
Publisher:W. H. Freeman

Fundamentals of Biochemistry: Life at the Molecul...
Biochemistry
ISBN:9781118918401
Author:Donald Voet, Judith G. Voet, Charlotte W. Pratt
Publisher:WILEY

Biochemistry
Biochemistry
ISBN:9781305961135
Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougal
Publisher:Cengage Learning

Biochemistry
Biochemistry
ISBN:9781305577206
Author:Reginald H. Garrett, Charles M. Grisham
Publisher:Cengage Learning

Fundamentals of General, Organic, and Biological ...
Biochemistry
ISBN:9780134015187
Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. Peterson
Publisher:PEARSON
Anaerobic Respiration; Author: Bozeman Science;https://www.youtube.com/watch?v=cDC29iBxb3w;License: Standard YouTube License, CC-BY