
Concept explainers
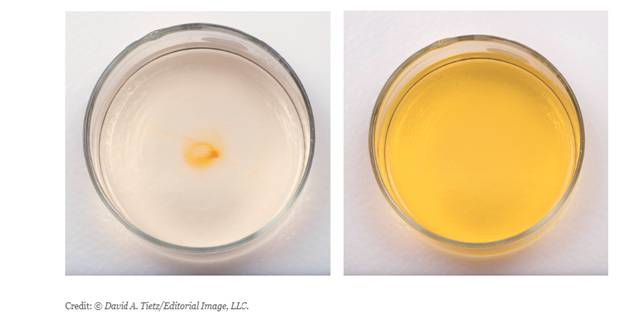
Iodine
Describe the change in solubility of I2 in terms of the change in intermolecular forces.
Want to see the full answer?
Check out a sample textbook solution
Chapter 13 Solutions
Chemistry
- In a solution with carbon tetrachloride as the solvent, the compound VCl4. undergoes dimerization: 2VCl4V2Cl8 When 6.6834 g VCl4. is dissolved in 100.0 g carbon tetrachloride, the freezing point is lowered by 5.97C. Calculate the value of the equilibrium constant for the dimerization of VCl4 at this temperature. (The density of the equilibrium mixture is 1.696 g/cm3, and Kf = 29.8C kg/mol for CCl4.)arrow_forwardSolubility is an equilibrium position, whereas Ksp is an equilibrium constant. Explain the difference.arrow_forwardAt some specific temperature, silver bromide, AgBr(s), has a measured solubility of 6.43×10-7 M. Calculate the Ksp value for AgBr(s) at this temperature.arrow_forward
- Calcium hydroxide is a base that does not dissolve very well in water. When saturated, a calcium hydroxide solution at 25°C contains 1,3 gram Ca(OH)2 per dm3 solution. The molar mass of Ca(OH)2 is 74,1 g×mol–1. When the Ca(OH)2 dissolves, the following reaction represents the solubility equilibrium: Ca(OH)2 (s) D Ca2+ (aq) + 2 OH– (aq) Use this information above to find the value of Ksp for Ca(OH)2 at 25°C.arrow_forwardYou have a solution of 0.0010 M AgNO3 (silver nitrate; nitrate salts are very soluble in water). To this you add AgBr. What is the molar solubility of AgBr in the solution? The Ksp for silver bromide (AgBr) in water is 5.35 x 10-13 at 25 °C.arrow_forwardAt 25 °C the molar solubility of nickel(II) carbonate (NiCO3, Ksp = 1.42 x 10-7) in pure water is 3.77 x 10-4 M. How will the molar solubility of NiCO3 change (increase, decrease, stay the same, not enough information) in a solution that contains 0.1 M sodium carbonate (Na2CO3)?arrow_forward
- The solubility of silver phosphate, Ag3PO4 at 25°C is 1.61 x 10-5 mol/L. Determine the concentration of the Ag+ ion in a saturated solution.arrow_forwardWhat is the molar solubility (in M to two decimal places) of copper(II) hydroxide in water (at 25 °C)? Ksp = 2.2 × 10–20arrow_forwardThe molar solubility of Fe(OH)2 at 25 °C is 1.6×10−5. What is Ksp for Fe(OH)2? 1.0×10−9 1.6×10−2 1.6×10−14 4.1×10−15arrow_forward
- At 25 °C, for Lead (II) sulphate (PbSO4), Ksp = 2.53×10-8 and for Barium sulphate (BaSO4) Ksp = 1.08×10-10. Suppose that Na2SO4 is added gradually to 500 mL of a solution that contains both Pb2+ ion and Ba2+ ion. (i) What will the Ba2+ concentration be (in mol/l) when Pb just begins to precipitate if the initial solution contains 0.40 M Pb2+ and 0.52 M Ba2+? (ii) What percentage of the strontium ion has precipitated when PbSO4 just begins to precipitate if the initial solution contains 0.28 M Pb2+ and 0.32 M Ba2+?arrow_forwardThe molar solubility of the metal hydroxide (M(OH)2) in water is: M(OH)2 = M2+ + 20H Ksp = 5.02 x 1o-6 1.00 x 10-5 M O 4.31 x 10-6 M 3.30 x 10-6 M O 3.60 x 10-2 M O 1.10 x 10-2 Marrow_forwardThe solubility of manganese (II) hydroxide (Mn(OH)2) is 2.2 x 10-5 M. What is the Ksp of Mn(OH)2?arrow_forward
 Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning





