
Chemistry
4th Edition
ISBN: 9780078021527
Author: Julia Burdge
Publisher: McGraw-Hill Education
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 13.4, Problem 1PPC
Practice Problem CONCEPTUALIZE
CONCEPTUALIZE
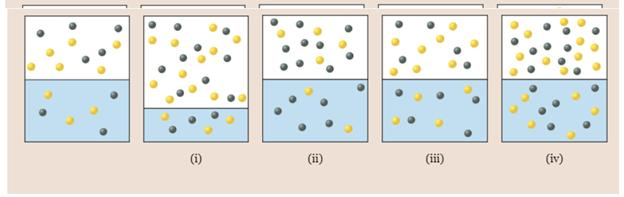
The first diagram represents a closed system with two different gases dissolved in water. Which of the other diagrams could represent a closed system consisting of the same two gases at the same temperature?
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
What is the freezing point of a solution prepared by dissolving 11.3 f of Ca(NO3)2 (molar mass = 164.1 g/mol) in 115 g of water? The freezing point depression constant for water is 1.86oC/m.
(A) -1.11oC
(B) -2.23oC
(C) -3.34oC
(D) 0.00oC
Creative Com In 300. g of water at 50 degrees Celsius, approximately what mass of substance C will dissolve?
Q1 0.1 MOLAL AQUEOUS SOLUTION OG Al2(SO4)3 is 70% dissociated.the vant hoff factor of the salt
(a)2.5
(b)4.2
(c)4.8
(d)3.8
Q2 which colligative property is widely used to determine molar masses of macromolecule
(a)osmotic pressure
(b)RLVP
(c)depression of freezing point
(d)elevation in boiling point
Q3 the pressure of H2 required to make the potential of H2 electrode zero in 1M HCl solution at 298K Is
(a)1atm
(b)10atm
(c)0.1atm
(d)0.01atm
Q4 the gas produced at cathode during the electrolysis of dilute H2SO4 solution is
(a)O2
(b)SO2
(c)SO3
(d)H2
Chapter 13 Solutions
Chemistry
Ch. 13.1 - Prob. 1PPACh. 13.1 - Prob. 1PPBCh. 13.1 - Prob. 1PPCCh. 13.2 - Prob. 1CPCh. 13.2 - Prob. 2CPCh. 13.2 - Practice Problem ATTEMPT
Determine (a) the...Ch. 13.2 - Practice Problem BUILD
Determine the molality of...Ch. 13.2 - Prob. 1PPCCh. 13.3 - Practice Problem ATTEMPT
An aqueous solution that...Ch. 13.3 - Practice Problem BUILD
Determine the percent...
Ch. 13.3 - Practice Problem CONCEPTUALIZE
The diagrams...Ch. 13.3 - Prob. 1CPCh. 13.3 - What is the molality of a solution prepared by...Ch. 13.3 - Prob. 3CPCh. 13.3 - Prob. 4CPCh. 13.4 - Practice ProblemATTEMPT Calculate the...Ch. 13.4 - Prob. 1PPBCh. 13.4 - Practice Problem CONCEPTUALIZE
The first diagram...Ch. 13.4 - The solubility of N2 in water at 25°C and an N 2...Ch. 13.4 - Calculate the molar concentration of O 2 in water...Ch. 13.5 - Practice ProblemATTEMPT Calculate the vapor...Ch. 13.5 - Prob. 1PPBCh. 13.5 - Practice ProblemCONCEPTUALIZE The diagrams...Ch. 13.5 - 13.5.1 A solution contains 75.0 g of glucose...Ch. 13.5 - Determine the boiling point and the freezing point...Ch. 13.5 - 13.5.3 Calculate the osmotic pressure of a...Ch. 13.5 - 13.5.4 A 1.00-m solution of has a freezing point...Ch. 13.6 - Prob. 1PPACh. 13.6 - Prob. 1PPBCh. 13.6 - Practice Problem CONCEPTUALIZE
The diagrams...Ch. 13.6 - 13.6.1 A solution made by dissolving 14.2 g of...Ch. 13.6 - Prob. 2CPCh. 13.7 - Practice ProblemATTEMPT The freezing-point...Ch. 13.7 - Practice ProblemBUILD Using the experimental van't...Ch. 13.7 - Practice Problem CONCEPTUALIZE
The diagram...Ch. 13.8 - Practice ProblemATTEMPT Determine the osmotic...Ch. 13.8 - Practice Problem BUILD
Determine the...Ch. 13.8 - Practice Problem CONCEPTUALIZE
The first diagram...Ch. 13.9 - Practice Problem ATTEMPT
Calculate the molar mass...Ch. 13.9 - Practice Problem BUILD
What mass of naphthalene...Ch. 13.9 - Practice Problem CONCEPTUALIZE
The first diagram...Ch. 13.10 - Practice Problem ATTEMPT A solution made by...Ch. 13.10 - Practice Problem BUILD What mass of insulin must...Ch. 13.10 - Practice ProblemCONCEPTUALIZE The first diagram...Ch. 13.11 - Practice Problem ATTEMPT An aqueous solution that...Ch. 13.11 - Practice Problem BUILD
An aqueous solution that is...Ch. 13.11 - Practice Problem CONCEPTUALIZE The diagrams...Ch. 13 - Which of the following processes is accompanied by...Ch. 13 - 13.2
For each of the processes depicted here,...Ch. 13 - 13.3
For each of the processes depicted here,...Ch. 13 - Prob. 4KSPCh. 13 - Describe and give examples of an unsaturated...Ch. 13 - Prob. 2QPCh. 13 - Prob. 3QPCh. 13 - Prob. 4QPCh. 13 - Prob. 5QPCh. 13 - As you know, some solution processes are...Ch. 13 - Prob. 7QPCh. 13 - 13.8 Describe the factors that affect the...Ch. 13 - Prob. 9QPCh. 13 - Prob. 10QPCh. 13 - Prob. 11QPCh. 13 - Prob. 12QPCh. 13 - Prob. 13QPCh. 13 - Prob. 14QPCh. 13 - Prob. 15QPCh. 13 - Prob. 16QPCh. 13 - Prob. 17QPCh. 13 - Prob. 18QPCh. 13 - Prob. 19QPCh. 13 - Prob. 20QPCh. 13 - 13.21 The alcohol content of hard liquor is...Ch. 13 - Prob. 22QPCh. 13 - Prob. 23QPCh. 13 - 13.24 The density of an aqueous solution...Ch. 13 - Prob. 25QPCh. 13 - Prob. 26QPCh. 13 - Prob. 27QPCh. 13 - What is thermal pollution? Why is it harmful to...Ch. 13 - Prob. 29QPCh. 13 - A student is observing two beakers of water. One...Ch. 13 - Prob. 31QPCh. 13 - Prob. 32QPCh. 13 - The solubility of KNO 3 is 155 g per 100 g of...Ch. 13 - Prob. 34QPCh. 13 - 13.35 The solubility of in water at What is its...Ch. 13 - Prob. 36QPCh. 13 - Prob. 37QPCh. 13 - Prob. 38QPCh. 13 - Prob. 39QPCh. 13 - Prob. 40QPCh. 13 - Prob. 41QPCh. 13 - Prob. 42QPCh. 13 - Prob. 43QPCh. 13 - Prob. 44QPCh. 13 - Prob. 45QPCh. 13 - 13.46 Write the equations relating boiling-point...Ch. 13 - Prob. 47QPCh. 13 - Prob. 48QPCh. 13 - Prob. 49QPCh. 13 - Prob. 50QPCh. 13 - Prob. 51QPCh. 13 - Prob. 52QPCh. 13 - Prob. 53QPCh. 13 - What are ion pairs? What effect does ion-pair...Ch. 13 - Prob. 55QPCh. 13 - Prob. 56QPCh. 13 - 13.57 A solution is prepared by dissolving 396 g...Ch. 13 - Prob. 58QPCh. 13 - Prob. 59QPCh. 13 - Prob. 60QPCh. 13 - Prob. 61QPCh. 13 - Prob. 62QPCh. 13 - Prob. 63QPCh. 13 - 13.64 How many liters of the antifreeze ethylene...Ch. 13 - Prob. 65QPCh. 13 - Prob. 66QPCh. 13 - Prob. 67QPCh. 13 - Prob. 68QPCh. 13 - 13.69 Both and are used to melt ice on roads and...Ch. 13 - Prob. 70QPCh. 13 - Prob. 71QPCh. 13 - Prob. 72QPCh. 13 - Prob. 73QPCh. 13 - Calculate the difference in osmotic pressure (in...Ch. 13 - 13.75 Which of the following aqueous solutions has...Ch. 13 - Prob. 76QPCh. 13 - 13.77 Arrange the following solutions in order of...Ch. 13 - Prob. 78QPCh. 13 - Indicate which compound in each of the following...Ch. 13 - Prob. 80QPCh. 13 - Prob. 81QPCh. 13 - Prob. 82QPCh. 13 - Prob. 83QPCh. 13 - The elemental analysis of an organic solid...Ch. 13 - 13.85 A solution of 2.50 g of a compound having...Ch. 13 - 13.86 The molar mass of benzoic acid determined...Ch. 13 - 13.87 A solution containing 0.8330 g of a polymer...Ch. 13 - Prob. 88QPCh. 13 - A solution of 6.85 g of a carbohydrate in 100.0 g...Ch. 13 - Prob. 90QPCh. 13 - Prob. 91QPCh. 13 - Prob. 92QPCh. 13 - Prob. 93QPCh. 13 - Prob. 94QPCh. 13 - Prob. 95APCh. 13 - Prob. 96APCh. 13 - 13.97 Acetic acid is a polar molecule and can form...Ch. 13 - Prob. 98APCh. 13 - Prob. 99APCh. 13 - Prob. 100APCh. 13 - Prob. 101APCh. 13 - Prob. 102APCh. 13 - Prob. 103APCh. 13 - Prob. 104APCh. 13 - Prob. 105APCh. 13 - A solution of 1.00 g of anhydrous aluminum...Ch. 13 - Explain why reverse osmosis is (theoretically)...Ch. 13 - A 1.32-g sample of a mixture of cyclohexane ( C 6...Ch. 13 - Prob. 109APCh. 13 - Prob. 110APCh. 13 - Prob. 111APCh. 13 - Prob. 112APCh. 13 - Prob. 113APCh. 13 - Prob. 114APCh. 13 - Prob. 115APCh. 13 - Iodine ( I 2 ) is only sparingly soluble in water...Ch. 13 - Concentrated hydrochloric acid is usually...Ch. 13 - Explain each of the following statements: (a) The...Ch. 13 - A mixture of NaCl and sucrose ( C 12 H 22 O 12 )...Ch. 13 - Prob. 120APCh. 13 - At 27°C, the vapor pressure of pure water is 23.76...Ch. 13 - A nonvolatile organic compound Z was used to make...Ch. 13 - Prob. 123APCh. 13 - Prob. 124APCh. 13 - Prob. 125APCh. 13 - Prob. 126APCh. 13 - Prob. 127APCh. 13 - Prob. 128APCh. 13 - Prob. 129APCh. 13 - Prob. 130APCh. 13 - Prob. 131APCh. 13 - Consider the three mercury manometers shown in the...Ch. 13 - Prob. 133APCh. 13 - Prob. 134APCh. 13 - Prob. 135APCh. 13 - 13.136 In the apparatus shown, what will happen if...Ch. 13 - Prob. 137APCh. 13 - Prob. 138APCh. 13 - Lysozyme is an enzyme that cleaves bacterial cell...Ch. 13 - Prob. 140APCh. 13 - Prob. 141APCh. 13 - Prob. 142APCh. 13 - Prob. 143APCh. 13 - Prob. 144APCh. 13 - Prob. 145APCh. 13 - What masses of sodium chloride, magnesium...Ch. 13 - Prob. 147APCh. 13 - Prob. 148APCh. 13 - Prob. 149APCh. 13 - Hemoglobin, the oxygen-transport protein, binds...Ch. 13 - Prob. 151APCh. 13 - 13.152 The vapor pressure of ethanol and the...Ch. 13 - Prob. 153APCh. 13 - A mixture of two volatile liquids is said to be...Ch. 13 - A mixture of two volatile liquids is said to be...Ch. 13 - Prob. 3SEPPCh. 13 - Prob. 4SEPP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Determine whether the following solutions will be saturated at temperature of 20°C. (A) 24.9 g of potassium chloride dissolved in 100 g of water (B) 44.0 g of sodium nitrate dissolved in 50.0 g of water (C) 29.6 g of potassium chlorate dissolved in 400 g of water (D) 115 g of surcose C6H22O11, dissolved in 115 g of waterarrow_forwardWhat would be the theoretical freezing point of a solution made by dissolving 3.313 g of KCl in 135.0 g of water? The freezing point constant of water is 1.86 °C/molal. ( I am studying for my exam this is a practice problem but the answer I got its there.I got -1.225 -0.8368 °C 0.8368 °C 1.195 °C -1.016 °C -1.195 °Carrow_forward1-If 5.06 g of KNO3 is dissolved in 10.00 g water at 31 C°, then the solubility of KNO3in water is (а) 50.6 mlb) 0.506 (c) 197.62 (d) 1.98arrow_forward
- A solution containing 25.0% by mass of ammonium sulfate, (NH4)2SO4 (molar mass = 132.14 g/mol) has a density of 1.15 g/mL. What is the molality of the solution? (molal (m) = mol/kg of solvent) (A) 1.64 m (B) 1.89 m (C) 2.18 m (D) 2.52 marrow_forward17) Which response lists all the following pairs that are miscible liquids? Pair # 1. octane (C8H18) and water Pair # 2. ammonia (NH3) and water Pair # 3. octane (C8H18) and carbon tetrachloride (CC14) B) 2,3 A) 2 C) 3 D) 1, 2 E) 1,3 17)arrow_forwardView Policies Show Attempt History Current Attempt in Progress What masses of nitrogen and oxygen are dissolved in 159 g of water at 20.0 °C when the water is saturated with air, in which PN2 equals 593 torr and Poz equals 159 torr? At 1.00 atm pressure, the solubility of pure oxygen in water is 0.00430 g O2/100.0 g H2O, and the solubility of pure nitrogen in water is O.00190 g N/100.0 g HO. mg of nitrogen mg of oxygen eTextbook and Media Hint Save for Later Attempts: 1 of 3 used Submit Answerarrow_forward
- 8. At a pressure of 1 atm, a solution of sucrose (C12H22O11) in water has a boiling point of 101 °C. If the Kb of water is 0.5 °C/m, then the concentration of the solution is... (A) 5.0 M (B) 4.0 M (C) 2.5 M (D) 2.0 M (E) 1.0 Marrow_forward9. A certain drug with molecular weight, M = 110.5 g/mol, has a surface tension y = 8.2 x 10³ erg/cm² and density p = 1.1 g/cm³. What particle size is required to increase the solubility by 5% at 25°C? (R= 8.314 x 107 erg/K mol) (a) 18.4 µm (b) 3.7 μm (c) 7.8 μm (d) 13.6 μm 0.5.1arrow_forward6.35 If 68.4 g of sucrose (M 342 g mol-¹) is dissolved in 1000 g of water, (a) what is the vapor pressure at 20 °C? (b) What is the freezing point? The vapor pressure of water at 20°C is 2.3149 kPa. =arrow_forward
- 3:47 PM Mon Feb 6 Question 16 of 16 Calculate the vapor pressure of ethanol, CH3CH₂OH, in a water solution containing 82.5% ethanol by volume at 19.0 °C. The vapor pressure of pure ethanol at 19.0 °C is 40.0 Torr. The density of ethanol is 0.789 g.mL-¹ and the density of water is 1.000 g.mL-¹1. Tap here or pull up for additional resources.arrow_forwardpls answer thank you A mixture of ethanol (C2H5OH) and water has a freezing point of -16° C. (a) what is the molality of the solution? (b) What is the mass of the alcohol present?arrow_forward(C) 9.55e-02 atm (D) 2.30e-02 atm (E) The correct answer is not shown. How many grams of ethylene glycol (C2H602) must be added to 1.420 kg of water to produce a solution that freezes 9. at -8.60°C? (A) The correct answer is not shown. (B) 1.01e+03 g (C) 4.08e+02 g (D) 5.88e+02 g (E) 5.11e+02 g 11:57 AM O 4) P 3/8/2021 PrtScn Home End PgUp PgDn F11 Ins F7 F8 F9 F10 F12 8 9 { } (8) 近arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY
Solutions: Crash Course Chemistry #27; Author: Crash Course;https://www.youtube.com/watch?v=9h2f1Bjr0p4;License: Standard YouTube License, CC-BY