
Concept explainers
(a)
Interpretation:
The product obtained by the reaction of the given
Concept Introduction:
Hydrogenation reaction is nothing but the addition of hydrogen to an alkene. In this reaction two bonds are simultaneously broken, one is carbon-carbon double bond, and second one is hydrogen-hydrogen bond, finally forms two carbon-hydrogen bonds. The general reaction can be represented as follows,
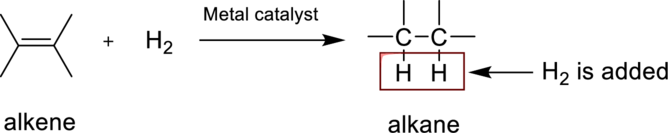
(b)
Interpretation:
The product obtained by the reaction of the given alkene with hydrogen and palladium catalyst has to be identified.
Concept Introduction:
Refer to part (a).
(c)
Interpretation:
The product obtained by the reaction of the given alkene with hydrogen and palladium catalyst has to be identified.
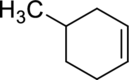
Concept Introduction:
Refer to part (a).
(d)
Interpretation:
The product obtained by the reaction of the given alkene with hydrogen and palladium catalyst has to be identified.
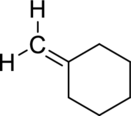
Concept Introduction:
Refer to part (a).
Want to see the full answer?
Check out a sample textbook solution
Chapter 11 Solutions
Principles of General, Organic, Biological Chemistry
- what type of reaction is exhibited when alkene is converted into alcohol? * O hydration O hydrogenation O hydrohalogenation halogenation The formula of anesthetic "ether" is * О снзоснз O C2H50C2H5 O C2H50H О снзсн2сосн2снзarrow_forwardWhat is the major difference between the hydration of an alkene and the hydration of an alkyne?arrow_forwardWhat alcohol is formed when the alkene CH3CH2CH=CH2 is treated with H2O in the presence of H2SO4?arrow_forward
- Which of the following pairs of cycloalkanes represent structural isomers? a. b. c. d.arrow_forwardWhat is the molecular formula for each of the following hydrocarbons? a. 5-carbon alkene with one double bond b. 5-carbon alkene with two double bonds c. 5-carbon cycloalkene with one double bond d. 5-carbon cycloalkene with two double bondsarrow_forwardWhat type of hybridized orbital is present on carbon atoms bonded by a double bond? How many of these hybrid orbitals are on each carbon atom?arrow_forward
- What type of hybridized orbital is present on carbon atoms bonded by a triple bond? How many of these hybrid orbitals are on each carbon atom?arrow_forward3 Give the systematic name of the given alkene based on its semi-structural formula. CH, CH, CH, CH сн, — с %3Dсн CH CH2 - CH2 CH3 CH, Systematic name:arrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning





