
Concept explainers
(a)
Interpretation:
Whether the given pair of compounds related as constitutional isomers, stereoisomers, or identical has to be indicated.
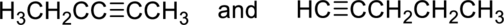
Concept Introduction:
Constitutional isomer is nothing but the structural isomer which has same molecular formula but differ in their connectivity of atoms.
Stereoisomers are spatial isomers which have same molecular formula and have differ in special arrangements which means the three-dimensional orientations of their atoms in space are arranged in different manner.
(b)
Interpretation:
Whether the given pair of compounds related as constitutional isomers, stereoisomers, or identical has to be indicated.
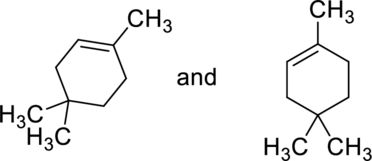
Concept Introduction:
Refer to part (a).
(c)
Interpretation:
Whether the given pair of compounds related as constitutional isomers, stereoisomers, or identical has to be indicated.
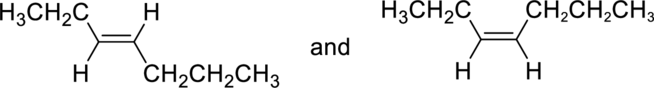
Concept Introduction:
Refer to part (a).
Want to see the full answer?
Check out a sample textbook solution
Chapter 11 Solutions
Principles of General, Organic, Biological Chemistry
- For each of the following compounds and ions,1. Draw a Lewis structure.2. Show the kinds of orbitals that overlap to form each bond.3. Give approximate bond angles around each atom except hydrogen.(a) [NH2]- (b) [CH2OH]+ (c) CH2“N¬CH3(d) CH3¬CH“CH2 (e) HC‚C¬CHO (f) H2N¬CH2¬CNarrow_forward1) Draw 3 constitutional isomers of C3H6O. 2) Which has the highest boiling point? Why? 3) Which one is most stable in water? Why?arrow_forwardDraw the structure of an alkane with molecular formula C7H16 that contains (a) one 4° carbon; (b) only 1° and 2° carbons; (c) 1°, 2°, and 3° hydrogens.arrow_forward
- Two compounds with the formula CH3 ¬ CH “ N ¬ CH3 are known Explain why only one compound with the formula (CH3)2CNCH3 is knownarrow_forward(b) Draw structural formulas of at least two alkynes of each molecular formula.(1) C6H10arrow_forwardDraw the five constitutional isomers having molecular formula C6H14.arrow_forward
- 2,3-Dibromoprop-1-ene (C3H4Br2) has four H atoms. Suppose that any of these H atoms can be replaced by a Cl atom to yield a molecule with the formula C3H3Br,Cl. (a) Identify two H atoms where this substitution would yield constitutional isomers of C3H3Br,CI; (b) enantiomers of C3H3Br,Cl; (c) diastereomers of C3H3Br,Cl. H нн H Br Br 2,3-Dibromoprop-1-enearrow_forwardDraw three constitutional isomers having molecular formula C7H14 that contains a five-membered ring and two methyl groups bonded to that ring.arrow_forwardWhich of the following line drawings represents the correct stereochemistry o molecule shown in the Newman projection below? E + Н. H I CH₂CH3 LOH H CH3 OH OH || IV OH 5.... OH A) I B) II C) III D) IV Karrow_forward
- 2.) Draw 1 isomer of the compounds in item no. 3, and state the type of isomerism. Example: 2-hydroxy-pentanal OH Structure: Isomer: он Type of isomerism: Functional Isomerismarrow_forwardDraw all constitutional isomers that have the molecular formula C4H6. (There are nine isomers.)arrow_forwarda) Draw the two ring flipped conformers of each molecule A and B. Indicate the more stable conformer, respectively. CH3 fCH3 H,C. CH3 H3C. CH3 CI CI A B a) Which is more stable between A and B? Why?arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





