
Concept explainers
(a)
Interpretation:
The given compound B to condensed structure and its IUPAC name has to be provided.
Concept Introduction:
There are about eight rules to be followed while giving IUPAC name for alkene.
- The suffix –ane has to be replaced with the suffix –ene. This is used to indicate the presence of double bond.
- The longest continuous chain of carbon atoms has to be chosen that contains both carbon atoms of the double bond.
- The parent carbon chain has to be numbered in a way so that the numbering begins at the end near to the double bond. In case if the double bond is equidistant from both ends, then numbering has to be done from the end that is closer to substituents.
- The position of the double bond has to be given a single number which is lower‑numbered carbon atom that is present in the double bond.
- Suffixes like –diene, -triene, -tetrene, and so on are used when the compound contains more than one double bond.
- In case of cycloalkenes which do not have any substitution, the numbering is not needed to locate the double bond because the bond is assumed to be between the carbons 1 and 2.
- In case if substituents are present in cycloalkene, then the double‑bonded carbon
atoms are numbered 1 and 2 in a direction where the substituent gets the lower number. - If the cycloalkenes contain more than one double bond, then one double bond is assigned the numbers 1 and 2 followed by the other double bond so that the lowest number possible is given.
(b)
Interpretation:
The product obtained by the reaction of the given alkene with hydrogen and metal catalyst has to be identified.
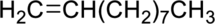
Concept Introduction:
Hydrogenation reaction is nothing but the addition of hydrogen to an alkene. In this reaction two bonds are simultaneously broken, one is carbon-carbon double bond, and second one is hydrogen-hydrogen bond, finally forms two carbon-hydrogen bonds. The general reaction can be represented as follows,
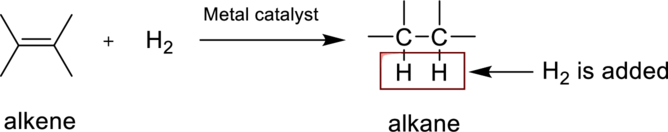
(c)
Interpretation:
The product obtained by the reaction of the given alkene with water in the presence of sulfuric acid has to be identified.
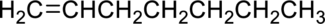
Concept Introduction:
Hydaration reaction is nothing but the addition of water to an alkene. In this reaction two bonds are simultaneously broken, one is carbon-carbon double bond, and second one is
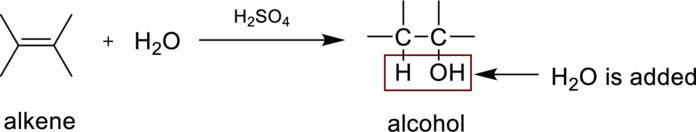
In the addition of water to an unsymmetrical alknene, the hydrogen from water is bonded to the less substituted carbon atom and is called Markovnikov’s rule.
Want to see the full answer?
Check out a sample textbook solution
Chapter 11 Solutions
Principles of General, Organic, Biological Chemistry
- 4 Answer the following questions about compound B, represented in the given ball-and-stick model. a. Convert B to a condensed structure and give its IUPAC name. b. What product is formed when B is treated with H 2 in the presence of a metal catalyst? c. What product is formed when B is treated with H 2O in the presence of H 2SO 4? d. What polymer is formed when B is polymerized?arrow_forward8. Give the IUPAC name for each alkene. CH3 CH2CH,CH3 Br а.arrow_forwardGive the IUPAC name for each alkene.arrow_forward
- 2. Complete the following reactions for the preparation of alcohols. Draw the structure of the product. Name the reactant and the product. a) CHy CH=CH-CHy + HyO Hydration of alkenes b) Cat. CHy-CHy + H2 drogenation carbonyl pups d)arrow_forwardDraw all alkenes that react with one equivalent of H2 in the presence of a palladium catalyst to form each alkane. Consider constitutional isomers only. a. b.arrow_forward1. Draw the following compounds. c. (Z)-2-methylpent-2-enol d. ethylpropanoatearrow_forward
- What is the structure of the IUPAC name? Draw the structure and give how many carbons are on the parent chain. (2Z)-3-sulfanylhex-2-en-4-ynedialarrow_forwardName each alkene: a. CH3CH=CHCH2CH2CH3 b. CH3CH=CH2 c. CH2CH3 l CH2=CHCH2CHCH3arrow_forwardAnswer each question using the ball-and-stick model of compound A. Draw a stereoisomer for A and give its IUPAC name.Draw a constitutional isomer that contains an OH group and give itsIUPAC name.arrow_forward
- What alkenes are formed when attached alcohol is dehydrated with TsOH?Label the major product when a mixture resultsarrow_forwardAcetophenone Condensed Structure: Product formed: A.Oxidation (K2Cr2O7/H2SO4) b.Reduction (NaBH4/H2O) c. Addition of alcohol ( CH3OH) → with another addition of alcohol (C2H5OH)arrow_forward• Whał arc the IUPAC namar of the ff. Carboxylic acias? a. COOH COOH CH3 b. I f. .COOH CHJCH2 ÇHCHCOOH CH3 NO2 tON C. COOH ноос d. COOHarrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

