
Concept explainers
(a)
Interpretation:
The given pair of
Concept Introduction:
Constitutional isomers are the isomer compounds which have the same molecular formula and different in connectivity. To predict whether two compounds are constitutional isomers, need to count the number of each atom in both compounds and look how the atoms are arranged.
Stereoisomers are the isomer compounds which differ only in special arrangements or three-dimensional arrangements of atoms.
(b)
Interpretation:
The given pair of alkenes as constitutional isomers or stereoisomers has to be labeled.
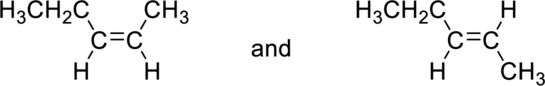
Concept Introduction:
Refer to part (a).
Want to see the full answer?
Check out a sample textbook solution
Chapter 11 Solutions
Principles of General, Organic, Biological Chemistry
- a. What hydrocarbon with molecular formula C4H10 forms only two monochlorinated products? Both products are achiral. b. What hydrocarbon with the same molecular formula as in part a forms three monochlorinated products? One is achiral and two are chiral.arrow_forwardFor which alkenes are stereoisomers possible? a. b. CH,CH,CH=CHCH3 C. -CH CHarrow_forwarda. What alkane, with molecular formula C5H12, forms only one monochlorinated product when it is heated with Cl2? b. What alkane, with molecular formula C7H16, forms seven monochlorinated products (disregarding stereoisomers) when heated with Cl2?arrow_forward
- Name each alkene. a. CH;=CH-CH;-CH3 CH, CH3 b. CH3-CH-c=CH-CH3 c. CH,=HC-CH–CH,-CH,-CH, CH3-CH ČH3 CH3 d. CH3-CH-CH=C-CH3 CH,-CH3arrow_forwardName each compound in which the benzene ring is best treated as a substituent. to CH3 a. CH,-CH-CH2-CH-CH2-ĊH-CH2-CH, CH2-CH, b. CH,-CH-CH=CH-CH,-CH,-CH,-CH,arrow_forwardDraw all constitutional isomers formed when each alkene is treated with NBS + hv. -CH3 CH c. CH2=C(CH2CH3)2 a. CH,CH=CHCH3 b.arrow_forward
- Rank each group of alkanes in order of increasing boiling point. Explain your choice of order. a. CH3CH2CH2CH2CH3, CH3CH2CH;CH3, CH3CH2CH3 b. CH;CH,CH,CH(CH)2, CH3(CH2),CH3, (CH3)2CHCH(CH3)karrow_forwardWhich projection corresponds to 2,3,4-trimethylhexane? CH,CH3 H3C. CH(CH3)2 ČH3 b. CH3 H CH,CH3 H. ČH3 (CH3)2CH CH3 H3C, C. HY CH;CH(CH3)2 ĊH3 CH3 d. (CH3),CH, H Y CH,CH3 ČH3 a.arrow_forwardProvide the IUPAC names for the following hydrocarbons. Do NOT consider stereochemistry. A. CH3 C=CC=CC=CC=CC=CCH=CH2 В. СH3 CH-CНCH-CHCH-СНCH-CH2arrow_forward
- Draw the products of combustion of each alkane. a. CH;CH,CH,CH2CH(CH3)2 b.arrow_forwardProvide the IUPAC names for the following hydrocarbons. Do NOT consider stereochemistry. A. B.CH, CH=CHC=CCH=CHCH-CHCH=CH₂| CH₂CH=CHC=CCH=CHC=CHarrow_forwardDraw the structure of the trans and cis alkene for those alkenes below that can exist as geometric isomers: i. CH:CH-CH-CН2 ii. CH3CH=CHCH3 iii. СH:CH2CH2CН-CH-CH-СHCH2CH2CH2CH2CHЗ iv. CH3CH2CH2CH2CH2CHCHCH2CH2CH2CH=CH2arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





